Research Article
Ambika Poudel
Ambika Poudel
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA.
Prabodh Satyal
Prabodh Satyal
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA.
Kathy Swor
Kathy Swor
Independent Researcher, 1432 W.
Heartland Dr., Kuna, ID 83634, USA.
William N. Setzer
William N. Setzer
Corresponding author
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA.
And
Department
of Chemistry, University of Alabama in Huntsville, Huntsville, AL 35899, USA.
E-mail: setzerw@uah.edu, wsetzer@chemistry.uah.edu; Tel.: +1-256-468-2862
Abstract
Achillea
millefolium is native to temperate regions of the
northern hemisphere and is important in traditional herbal medicines throughout
its range. The purpose of this work is to evaluate the essential oil
composition of the North American variety, A. millefolium var. occidentalis,
and to test the hypothesis that var. occidentalis has a chemical
composition different from Old World A. millefolium. Six samples of A.
millefolium were collected from different locations in the Great Basin
region of eastern Oregon and southern Idaho. The essential oils were obtained
by hydrodistillation and analyzed by gas chromatographic methods. The essential
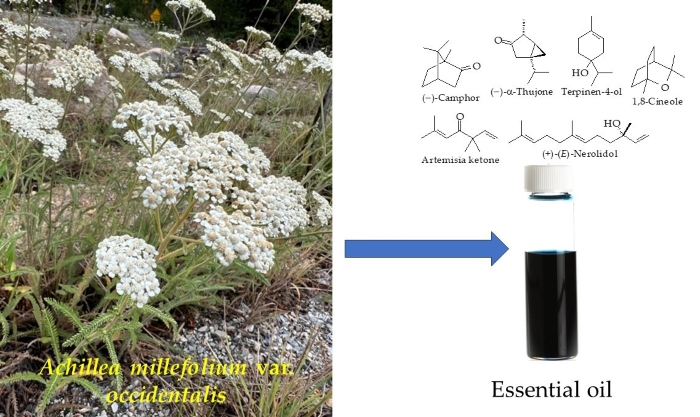
oils showed wide variation in composition with the major components (–)-camphor
(0.5-59.6%), (–)-α-thujone (0.0-40.5%), terpinen-4-ol (0.9-23.4%), artemisia
ketone (0.0-18.7%), 1,8-cineole (5.3-16.4%), and (+)-(E)-nerolidol
(0.4-13.5%). The essential oil compositions of western North American A.
millefolium cannot be distinguished from the samples from Old World samples
of A. millefolium; there is no phytochemical support for infraspecific
differentiation of the North American variety.
Abstract Keywords
Yarrow,
chemotaxonomy, gas chromatography, mass spectrometry, enantiomers.
1. Introduction
Achillea millefolium L. (Asteraceae) is a polymorphous species with a complex taxonomy [1, 2]. World Flora Online currently lists 161 synonyms for the species [3]. The plant is native to the temperate regions of the northern hemisphere, including Europe, western Asia, and North America [4], but has been introduced and cultivated worldwide. The plant is characterized by deeply divided pubescent leaves with flowers gathered in capitula, which are grouped in corymbs (Fig. 1). The western North American plant, known as Achillea millefolium var. occidentalis DC. (western yarrow), ranges from western Canada, south through the Dakotas, Colorado, and New Mexico, and into northern Mexico; and west to the Pacific Coast [5]. The Flora of North America, however, does not recognize infraspecific taxa of A. millefolium [6].
Figure 1. Achillea millefolium var. occidentalis. A: Photograph of plant
(by K. Swor). B: Scan of pressed plant (by W.N. Setzer).
Achillea millefolium has been used in traditional medicine throughout its global distribution [7]. The Shoshoni Native Americans applied a poultice of the boiled whole plant externally to treat sores, while a decoction of the plant was used as a liniment for sores and rashes. The Shoshoni prepared a decoction of the plant and took it to treat colds [8]. Similarly, the Paiute took an infusion of A. millefolium as a cough medicine [9]. In Europe, the plant is traditionally taken orally for gastrointestinal disorders or used topically to treat skin disorders [1]. In Iran, the plant is used for menstruation problems, to stimulate menstruation, to stop bleeding, for wound healing, to relieve asthma and constipation, and as diuretic, anthelmintic, and carminative [10]. In Colombia and Peru, A. millefolium is used to relieve gastrointestinal problems such as indigestion, flatulence, colitis, and gastritis [11]. The whole plant is used in Colombia to treat skin disorders, including acne, boils, botfly infestations, skin ulcers, and sores [11].
Achillea millefolium has been shown to be a source of sesquiterpene lactones, including 8-acetylegelolide, 8-angeloylegelolide, 8-angeloyloxyleucodin, acetylbalchanolide, achillicin, achillin, achillinin A, achillifolin, badkhyzin, millefin, millifolide C, isoachifolidiene, leukodin, dihydroparthenolide, and balchanolide; several polyacetylenes such as anacycline, dehydroanacycline, 2,9-pentadecadiene-12,14-diynoic acid isobutylamide, 2,4-tetradecadiene-8,10-diynoic acid 3-methylbutylamide, 2,4-undecadiene-8,10-diynoic acid 2,3-dehydropiperidide, and 2,4-undecadiene-8,10-diynoic acid isobutylamide; and flavonoids apigenin, artemetin, casticin, centauredidin, chrysoeriol, isorhamnetin, luteolin, and quercetin, as well as an azulene-containing essential oil [1, 7, 12, 13].
Although the taxonomic treatment in Flora of North America suggests little support for infraspecific taxa [6], it was deemed important to examine the essential oil compositions of several samples of A. millefolium collected in the wild from several locations in the Great Basin area of western North America to test the hypothesis that chemical composition differences may differentiate the A. millefolium var. occidentalis from Old World samples of A. millefolium.
2. Materials and methods
2.1. Plant material
Aerial parts of A. millefolium were collected from six individual plants growing in southern Idaho and eastern Oregon (Table 1). The plants were identified by W.N. Setzer in the field and verified by comparison with samples from the New York Botanical Garden [14]. Voucher specimens (WNS-Amo-5675 and WNS-Amo-7746) have been deposited at the University of Alabama in Huntsville herbarium. The fresh aerial parts were frozen and stored frozen (–20 °C) until processed.
2.2. Essential oil
For each sample of A. millefolium, the fresh/frozen aerial parts were finely chopped and hydrodistilled for three hours using a Likens-Nickerson apparatus [15–17] with continuous extraction of the distillate with dichloromethane to give the essential oils (Table 1).
Table 1. Collection and hydrodistillation details of Achillea millefolium var. occidentalis.
|
Sample Number |
Collection Date |
Collection Site |
Mass of Plant Material (g) |
Mass of Essential Oil (mg) |
Essential Oil Color |
|
#1 |
28 June 2022 |
Anderson Ranch, Idaho 43°24′20″N, 115°17′33″W, 1423 m asl |
73.21 |
236.2 |
deep blue |
|
#2 |
7 July 2022 |
Bogus Basin, Idaho 43°43′58″N, 116°7′42″W, 1673 m asl |
74.48 |
198.5 |
deep blue |
|
#3 |
27 May 2023 |
Leslie Gulch, Oregon 43°18’13”N, 117°17’17”W, 971 m asl |
98.25 |
1034.4 |
deep blue |
|
#4 |
8 August 2023 |
Redfish Lake, Idaho 44°8’40”N, 114°54’42”W, 2001 m asl |
83.14 |
145.9 |
yellow |
|
#5 |
11 August 2023 |
Silver City, Idaho 43°2’37”N, 116°46’33”W, 1788 m asl |
59.75 |
563.0 |
blue |
|
#6 |
26 August 2023 |
Boise Foothills, Idaho 43°41′20″N, 116°2′48″W, 1355 m asl |
32.76 |
475.7 |
deep blue |
2.3. Gas chromatographic analysis
The A. millefolium essential oils were analyzed by GC-MS, GC-FID, and enantioselective GC-MS as previously reported [18].
2.4. Hierarchical cluster analysis
Hierarchical cluster analysis (HCA) was carried out using XLSTAT v. 2018.1.1.62926 (Addinsoft, Paris, France). The concentrations of the 28 most abundant components (α-pinene, camphene, sabinene, β-pinene, yomogi alcohol, p-cymene, 1,8-cineole, artemisia ketone, γ-terpinene, cis-sabinene hydrate, terpinolene, α-thujone, β-thujone, camphor, borneol, terpinen-4-ol, α-terpineol, chrystanthenyl acetate, bornyl acetate, α-copaene, (E)-β-caryophyllene, germacrene D, (E)-nerolidol, spathulenol, caryophyllene oxide, γ-eudesmol, α-eudesmol, and chamazulene) from this study as well as previously reported compositions from the literature [19–52] and commercial samples from the Aromatic Plant Research Center (APRC) collection were used for the analysis. Dissimilarity was used to determine clusters considering Euclidean distance and Ward’s method was used to define agglomeration.
3. Results and discussion
Hydrodistillation of A. millefolium samples #1-#6 gave essential oils in yields of 0.323%, 0.267%, 1.053%, 0.175%, 0.942%, and 1.452%, respectively. Samples #1 and #2 showed the lowest yields; sample #6 showed the highest yield. It is not known what factors may be responsible for the yield variations; the samples were collected in different years, however, with the 2022 samples showing lower yields than the 2023 samples. The gas chromatographic analysis allowed the determination of a total of 152 compounds, which accounted for 95.0-99.1% of the compositions (Table 2).
Table 2. Chemical compositions (percent of total) of Achillea millefolium var. occidentalis essential oils.
|
RIcalc |
RIdb |
Compounds |
#1 |
#2 |
#3 |
#4 |
#5 |
#6 |
|
847 |
846 |
(Z)-Salvene |
- |
- |
0.1 |
- |
- |
- |
|
874 |
873 |
2-Methylbutyl acetate |
- |
- |
0.3 |
- |
- |
- |
|
902 |
902 |
Santolina triene |
- |
0.1 |
0.3 |
- |
0.1 |
- |
|
922 |
922 |
Artemisia triene |
- |
- |
0.2 |
tr |
tr |
- |
|
923 |
923 |
Tricyclene |
tr |
tr |
- |
tr |
0.1 |
0.3 |
|
925 |
927 |
α-Thujene |
tr |
tr |
0.2 |
tr |
0.2 |
0.1 |
|
932 |
932 |
α-Pinene |
0.3 |
0.4 |
0.9 |
0.2 |
1.4 |
2.2 |
|
945 |
942 |
5,5-Dimethylbut-3-enolide |
tr |
tr |
- |
- |
0.1 |
- |
|
949 |
950 |
Camphene |
0.4 |
1.0 |
0.1 |
0.4 |
1.7 |
7.3 |
|
960 |
959 |
Benzaldehyde |
tr |
tr |
0.1 |
0.1 |
0.1 |
tr |
|
969 |
967 |
Isoamyl propionate |
- |
tr |
0.1 |
- |
tr |
tr |
|
972 |
972 |
Sabinene |
3.2 |
1.1 |
3.1 |
0.8 |
2.3 |
0.7 |
|
977 |
978 |
β-Pinene |
0.5 |
0.5 |
2.2 |
0.3 |
6.3 |
1.6 |
|
988 |
989 |
Myrcene |
0.1 |
0.1 |
0.2 |
tr |
0.1 |
0.1 |
|
989 |
990 |
Dehydro-1,8-cineole |
0.1 |
tr |
0.1 |
0.1 |
0.2 |
0.1 |
|
995 |
996 |
Yomogi alcohol |
- |
1.0 |
8.8 |
2.1 |
3.6 |
tr |
|
1015 |
1015 |
Isoamyl isobutyrate |
0.1 |
0.1 |
0.3 |
0.1 |
0.1 |
0.1 |
|
1017 |
1017 |
α-Terpinene |
0.2 |
0.2 |
1.9 |
1.1 |
1.5 |
0.1 |
|
1024 |
1025 |
p-Cymene |
0.1 |
0.5 |
2.6 |
1.1 |
4.3 |
0.7 |
|
1028 |
1026 |
2-Acetyl-3-methylfuran |
- |
- |
- |
0.1 |
tr |
tr |
|
1029 |
1030 |
Limonene |
0.2 |
0.4 |
0.3 |
0.1 |
1.1 |
0.5 |
|
1030 |
1031 |
β-Phellandrene |
tr |
tr |
0.1 |
tr |
0.1 |
tr |
|
1032 |
1032 |
1,8-Cineole |
10.8 |
7.5 |
7.6 |
16.4 |
10.7 |
5.3 |
|
1035 |
1036 |
Benzyl alcohol |
- |
- |
0.1 |
0.2 |
- |
- |
|
1037 |
1034 |
Lavender lactone |
- |
- |
- |
- |
0.1 |
tr |
|
1042 |
1043 |
Phenylacetaldehyde |
0.1 |
0.1 |
tr |
0.1 |
0.2 |
0.2 |
|
1058 |
1056 |
Artemisia Ketone |
- |
- |
4.2 |
5.7 |
18.7 |
- |
|
1059 |
1058 |
γ-Terpinene |
0.5 |
1.9 |
4.2 |
1.4 |
- |
0.4 |
|
1069 |
1069 |
cis-Sabinene hydrate |
0.7 |
1.9 |
1.1 |
4.4 |
1.0 |
1.4 |
|
1079 |
1079 |
Artemisia alcohol |
- |
2.0 |
3.8 |
0.2 |
1.0 |
- |
|
1085 |
1086 |
Terpinolene |
0.1 |
0.1 |
0.7 |
0.8 |
0.6 |
0.1 |
|
1095 |
1096 |
Methyl benzoate |
- |
- |
0.1 |
0.2 |
- |
- |
|
1100 |
1101 |
Linalool |
0.8 |
1.0 |
- |
- |
0.1 |
tr |
|
1101 |
1101 |
trans-Sabinene hydrate |
- |
0.3 |
0.7 |
2.2 |
0.6 |
0.5 |
|
1103 |
1103 |
2-Methylbutyl isovalerate |
- |
0.8 |
1.1 |
- |
0.5 |
0.5 |
|
1103 |
1104 |
Hotrienol |
- |
- |
- |
- |
0.3 |
0.1 |
|
1105 |
1104 |
Nonanal |
- |
- |
- |
0.1 |
0.1 |
tr |
|
1106 |
1105 |
α-Thujone |
40.5 |
0.1 |
15.0 |
- |
- |
- |
|
1108 |
1103 |
2-Methylbutyl isovalerate |
- |
- |
- |
0.1 |
tr |
tr |
|
1112 |
1111 |
Phenethyl alcohol |
0.1 |
0.2 |
0.2 |
- |
- |
- |
|
1118 |
1118 |
β-Thujone |
5.6 |
tr |
1.8 |
- |
- |
- |
|
1120 |
1118 |
Dehydrosabina ketone |
- |
- |
- |
0.1 |
tr |
- |
|
1121 |
1122 |
trans-p-Mentha-2,8-dien-1-ol |
tr |
tr |
tr |
0.1 |
0.1 |
0.1 |
|
1124 |
1124 |
cis-p-Menth-2-en-1-ol |
0.1 |
0.1 |
0.5 |
1.3 |
0.5 |
0.1 |
|
1126 |
1126 |
α-Campholenal |
tr |
tr |
- |
0.1 |
0.1 |
0.2 |
|
1130 |
1131 |
Limona ketone |
tr |
- |
- |
tr |
tr |
- |
|
1136 |
1138 |
cis-p-Mentha-2,8-dien-1-ol |
tr |
- |
- |
- |
0.1 |
0.1 |
|
1139 |
1139 |
Nopinone |
- |
- |
- |
- |
0.1 |
- |
|
1140 |
1140 |
trans-Sabinol |
0.3 |
- |
0.1 |
- |
- |
- |
|
1141 |
1141 |
trans-Pinocarveol |
- |
- |
0.1 |
0.4 |
0.1 |
0.4 |
|
1142 |
1142 |
trans-p-Menth-2-en-1-ol |
0.1 |
0.1 |
0.3 |
1.0 |
0.4 |
tr |
|
1148 |
1149 |
Camphor |
7.2 |
19.4 |
0.5 |
15.8 |
8.8 |
59.6 |
|
1155 |
n.a. |
3,3,6-Trimethyl-4,5-heptadien-2-one |
- |
- |
- |
0.2 |
- |
- |
|
1156 |
1155 |
trans-Chrysanthemol |
- |
0.9 |
- |
- |
0.1 |
- |
|
1157 |
1157 |
Sabina ketone |
0.1 |
- |
- |
0.2 |
0.1 |
0.1 |
|
1159 |
1165 |
iso-Borneol |
0.1 |
- |
- |
- |
- |
- |
|
1160 |
1162 |
(Z)-iso-Citral |
- |
- |
- |
0.1 |
- |
- |
|
1162 |
1164 |
Pinocarvone |
0.1 |
tr |
- |
0.4 |
0.3 |
0.6 |
|
1164 |
1164 |
β-Artemisyl acetate |
0.1 |
1.6 |
3.8 |
0.9 |
1.5 |
- |
|
1169 |
1168 |
α-Phellandrene epoxide |
0.1 |
- |
- |
- |
- |
- |
|
1170 |
1170 |
δ-Terpineol |
0.2 |
- |
0.1 |
0.5 |
0.1 |
0.1 |
|
1172 |
1173 |
Borneol |
0.6 |
3.5 |
- |
0.3 |
0.7 |
1.3 |
|
1181 |
1180 |
Terpinen-4-ol |
1.7 |
2.4 |
9.0 |
23.4 |
8.9 |
0.9 |
|
1183 |
1183 |
Thuj-3-en-10-al |
0.1 |
- |
- |
- |
0.1 |
tr |
|
1187 |
1186 |
p-Cymen-8-ol |
tr |
tr |
tr |
0.1 |
0.2 |
0.1 |
|
1188 |
1191 |
1-Dodecene |
0.1 |
- |
- |
- |
- |
- |
|
1195 |
1195 |
α-Terpineol |
2.5 |
2.9 |
1.1 |
1.8 |
0.8 |
0.3 |
|
1196 |
1196 |
Myrtenal |
- |
- |
- |
- |
0.9 |
0.7 |
|
1198 |
1187 |
Cryptone |
0.2 |
0.1 |
- |
- |
0.2 |
0.1 |
|
1198 |
1196 |
cis-Piperitol |
- |
- |
0.1 |
0.1 |
- |
- |
|
1207 |
1208 |
Verbenone |
- |
- |
- |
- |
- |
0.1 |
|
1208 |
1207 |
(3E)-Octenyl acetate |
0.1 |
- |
- |
- |
- |
- |
|
1208 |
1208 |
trans-Piperitol |
0.1 |
tr |
0.2 |
- |
- |
- |
|
1218 |
1217 |
7-Ethylidene-bicyclo[3.3.0]octan-3-one |
0.3 |
- |
- |
0.1 |
- |
- |
|
1237 |
1237 |
Pulegone |
- |
- |
- |
- |
0.1 |
- |
|
1240 |
1240 |
Ascaridole |
- |
- |
- |
0.1 |
- |
- |
|
1241 |
1242 |
Cuminal |
0.1 |
- |
- |
0.1 |
0.1 |
- |
|
1243 |
1242 |
Carvone |
- |
- |
- |
- |
0.1 |
0.1 |
|
1269 |
1264 |
2,5-Bornanedione |
- |
- |
- |
- |
- |
0.1 |
|
1272 |
1266 |
Chrystanthemyl acetate |
- |
3.4 |
0.1 |
0.1 |
0.1 |
0.6 |
|
1283 |
1284 |
Lavandulyl acetate |
- |
- |
0.4 |
0.1 |
0.1 |
- |
|
1284 |
1285 |
Bornyl acetate |
0.6 |
5.4 |
0.1 |
0.7 |
1.7 |
2.2 |
|
1287 |
1286 |
trans-Sabinyl acetate |
0.3 |
- |
0.3 |
- |
- |
- |
|
1290 |
1291 |
p-Cymen-7-ol |
0.1 |
0.1 |
0.1 |
- |
- |
- |
|
1296 |
1296 |
Terpin-1-en-4-yl acetate |
- |
- |
0.1 |
0.1 |
0.2 |
- |
|
1298 |
1300 |
Carvacrol |
- |
tr |
0.1 |
0.1 |
0.1 |
tr |
|
1304 |
1306 |
iso-Ascaridole |
- |
- |
- |
0.1 |
tr |
- |
|
1327 |
1327 |
p-Mentha-1,4-dien-7-ol |
0.2 |
tr |
- |
- |
- |
- |
|
1334 |
1335 |
δ-Elemene |
0.1 |
- |
- |
- |
- |
- |
|
1352 |
1356 |
Eugenol |
0.4 |
0.6 |
0.2 |
0.3 |
0.2 |
0.1 |
|
1356 |
1349 |
α-Terpinyl acetate |
0.1 |
- |
- |
- |
- |
- |
|
1358 |
1361 |
Neryl acetate |
- |
0.1 |
- |
- |
- |
- |
|
1367 |
1367 |
Cyclosativene |
- |
0.1 |
- |
- |
- |
- |
|
1374 |
1375 |
α-Copaene |
- |
0.2 |
0.2 |
0.1 |
- |
0.1 |
|
1377 |
1378 |
Geranyl acetate |
- |
0.3 |
- |
- |
- |
- |
|
1383 |
1385 |
(E)-Jasmone |
0.1 |
0.1 |
- |
0.1 |
- |
- |
|
1384 |
1384 |
Benzyl pentanoate |
0.1 |
0.2 |
0.1 |
tr |
- |
- |
|
1386 |
1385 |
α-Isocomene |
0.1 |
- |
- |
- |
- |
0.1 |
|
1388 |
1390 |
trans-β-Elemene |
tr |
0.1 |
- |
- |
- |
tr |
|
1391 |
1392 |
(Z)-Jasmone |
0.2 |
1.0 |
0.2 |
0.2 |
0.1 |
tr |
|
1396 |
1396 |
(2E)-1,3,7-Trimethyl-2,6-octadienyl
acetate |
- |
0.4 |
- |
- |
0.1 |
- |
|
1410 |
1413 |
β-Isocomene |
0.1 |
- |
- |
- |
- |
- |
|
1419 |
1417 |
(E)-β-Caryophyllene |
0.6 |
1.7 |
0.5 |
0.2 |
0.5 |
0.5 |
|
1453 |
1452 |
(E)-β-Farnesene |
0.1 |
0.1 |
0.2 |
0.1 |
0.1 |
0.1 |
|
1455 |
1454 |
α-Humulene |
0.1 |
0.3 |
0.1 |
tr |
0.1 |
0.1 |
|
1481 |
1480 |
Germacrene D |
2.0 |
2.6 |
3.2 |
1.8 |
1.6 |
1.5 |
|
1483 |
1483 |
Davana ether 1 |
0.1 |
0.1 |
- |
- |
- |
- |
|
1484 |
1483 |
Phenethyl 2-methylbutyrate |
- |
0.1 |
0.1 |
- |
0.1 |
0.1 |
|
1488 |
1489 |
β-Selinene |
- |
- |
- |
- |
- |
0.1 |
|
1491 |
1489 |
(Z,E)-α-Farnesene |
- |
0.1 |
- |
- |
- |
- |
|
1494 |
1497 |
Bicyclogermacrene |
0.1 |
0.1 |
0.2 |
0.1 |
0.1 |
0.1 |
|
1494 |
1497 |
α-Selinene |
- |
- |
- |
- |
- |
0.1 |
|
1498 |
1497 |
α-Muurolene |
- |
0.3 |
0.2 |
tr |
- |
0.1 |
|
1502 |
1502 |
Davana ether 2 |
0.3 |
0.2 |
- |
- |
- |
- |
|
1504 |
1504 |
Davana ether 3 |
0.1 |
- |
- |
- |
- |
- |
|
1504 |
1503 |
(E,E)-α-Farnesene |
tr |
0.1 |
0.2 |
- |
tr |
- |
|
1512 |
1512 |
γ-Cadinene |
tr |
tr |
tr |
- |
- |
0.1 |
|
1514 |
1515 |
Cubebol |
- |
- |
tr |
- |
- |
0.1 |
|
1517 |
1518 |
δ-Cadinene |
0.1 |
0.2 |
0.1 |
0.1 |
0.1 |
0.2 |
|
1521 |
1521 |
Davana ether 4 |
0.2 |
0.1 |
- |
- |
- |
- |
|
1523 |
1523 |
β-Sesquiphellandrene |
0.1 |
- |
0.1 |
- |
- |
- |
|
1547 |
1549 |
α-Elemol |
0.1 |
- |
0.4 |
0.5 |
- |
- |
|
1560 |
1561 |
(E)-Nerolidol |
5.7 |
13.5 |
4.4 |
0.4 |
1.4 |
0.5 |
|
1569 |
1570 |
Neryl 2-methylbutanoate |
0.3 |
- |
0.1 |
tr |
0.1 |
- |
|
1572 |
1569 |
Longipinocarvone |
0.2 |
0.2 |
- |
0.3 |
0.5 |
0.3 |
|
1577 |
1580 |
Neryl isovalerate |
0.3 |
- |
0.3 |
0.1 |
0.1 |
0.2 |
|
1581 |
1587 |
Caryophyllene oxide |
0.7 |
0.8 |
0.5 |
0.5 |
1.3 |
1.0 |
|
1591 |
1593 |
Salvial-4(14)-en-1-one |
- |
- |
- |
0.1 |
0.1 |
- |
|
1597 |
1597 |
Fokienol |
- |
0.8 |
0.8 |
0.1 |
0.7 |
0.2 |
|
1608 |
1613 |
Copaborneol |
- |
0.3 |
0.2 |
- |
- |
- |
|
1610 |
1613 |
Humulene epoxide II |
- |
- |
- |
- |
tr |
tr |
|
1612 |
1609 |
Rosifoliol |
- |
- |
0.2 |
0.1 |
- |
- |
|
1621 |
1632 |
Humulenol II |
- |
- |
- |
0.5 |
1.7 |
0.7 |
|
1629 |
1627 |
Eremoligenol |
- |
- |
0.4 |
0.1 |
- |
- |
|
1631 |
1632 |
γ-Eudesmol |
- |
- |
3.0 |
- |
- |
- |
|
1632 |
1630 |
Caryophylla-4(12),8(13)-dien-5α-ol |
0.2 |
0.3 |
0.3 |
0.2 |
0.8 |
0.5 |
|
1635 |
1635 |
Caryophylla-4(12),8(13)-dien-5β-ol |
0.5 |
0.4 |
0.4 |
0.6 |
0.8 |
0.4 |
|
1641 |
1643 |
τ-Cadinol |
- |
- |
- |
- |
- |
0.4 |
|
1642 |
1642 |
Methyl (Z)-jasmonate |
0.1 |
0.9 |
0.2 |
- |
0.2 |
0.1 |
|
1654 |
1655 |
α-Eudesmol |
- |
- |
0.7 |
3.0 |
- |
0.1 |
|
1655 |
1655 |
α-Cadinol |
0.4 |
0.3 |
- |
- |
- |
0.1 |
|
1671 |
1671 |
14-Hydroxy-9-epi-(E)-caryophyllene |
- |
- |
- |
0.5 |
- |
0.1 |
|
1680 |
1676 |
8-Hydroxyisobornyl isobutanoate |
- |
- |
- |
0.2 |
1.2 |
0.6 |
|
1684 |
1683 |
Germacra-4(15),5,10(14)-trien-1α-ol |
- |
- |
0.3 |
1.3 |
- |
- |
|
1716 |
1716 |
(2E,6E)-Farnesol |
- |
0.1 |
0.2 |
0.1 |
- |
- |
|
1730 |
1730 |
Chamazulene |
4.4 |
7.0 |
0.8 |
- |
0.7 |
0.8 |
|
1775 |
1784 |
Dehydrochamazulene |
- |
0.1 |
- |
- |
- |
- |
|
1832 |
1832 |
(2Z,6E)-Farnesyl acetate |
- |
0.1 |
- |
- |
- |
- |
|
1932 |
1938 |
Pellitorine |
- |
- |
- |
- |
- |
0.4 |
|
2300 |
2300 |
Tricosane |
0.1 |
0.1 |
- |
0.1 |
0.1 |
tr |
|
2500 |
2500 |
Pentacosane |
0.1 |
0.1 |
- |
0.1 |
0.1 |
0.1 |
|
2700 |
2700 |
Heptacosane |
0.1 |
0.1 |
- |
0.1 |
0.1 |
tr |
|
|
|
Compound Classes |
|
|
|
|
|
|
|
Monoterpene
hydrocarbons |
5.7 |
6.5 |
17.0 |
6.2 |
19.7 |
13.8 |
||
|
Oxygenated
monoterpenoids |
74.3 |
53.8 |
60.3 |
79.7 |
64.0 |
75.8 |
||
|
Sesquiterpene
hydrocarbons |
7.8 |
12.8 |
5.7 |
2.2 |
3.1 |
3.8 |
||
|
Oxygenated
sesquiterpenoids |
8.5 |
17.3 |
11.7 |
8.3 |
7.2 |
4.3 |
||
|
Benzenoid
aromatics |
0.6 |
1.1 |
0.8 |
0.8 |
0.5 |
0.4 |
||
|
Others |
1.1 |
3.5 |
2.3 |
1.0 |
1.7 |
1.1 |
||
|
Total
identified |
97.9 |
95.0 |
97.8 |
98.2 |
96.2 |
99.1 |
RIcalc = Retention index determined using a homologous series of n-alkanes on a ZB-5ms column [53]. RIdb = Reference retention
index from the databases [54–57]. tr = trace (< 0.05%). - = not detected. Major components are highlighted in bold.
The major components were camphor (0.5-59.6%, mean 18.5%), α-thujone (0.0-40.5%, mean 9.3%), terpinen-4-ol (0.9-23.4%, mean 7.7%), artemisia ketone (0.0-18.7%, mean 4.8%), 1,8-cineole (5.3-16.4%, mean 9.7%), (E)-nerolidol (0.4-13.5%, mean 4.3%), yomogi alcohol (0.0-8.8%, mean 2.6%), camphene (0.1-7.3%, mean 1.8%), chamazulene (0.0-7.0%, mean 2.3%), β-pinene (0.3-6.3%, mean 1.2%), β-thujone (0.0-5.6%, mean 1.2%), and bornyl acetate (0.1-5.4%, mean 1.8%). The A. millefolium essential oil compositions show little similarity. However, samples #1 and #3 were rich in α-thujone, samples #2 and #6 were rich in camphor, and samples #4 and #5 were rich in terpinen-4-ol. Essential oil samples #1, #2, #3, #5, and #6 were blue in color, owing to the presence of chamazulene. Essential oil sample #4, which lacked chamazulene, was yellow in color.
In order to compare the essential oil compositions of the Great Basin samples in this work with the chemical compositions of A. millefolium from Old World locations, a hierarchical cluster analysis was carried out based on the 28 most abundant components. The cluster analysis shows much variation in essential oil compositions (Fig. 2), but four major groupings are evident: (1) a germacrene D/sabinene group, (2) a β-pinene/caryophyllene oxide group, (3) a 1,8-cineole/camphor group, and (4) a chamazulene group. Great Basin samples #1, #2, #4, #5, and #6 fall into the 1,8-cineole/camphor group, while sample #3 falls into the β-pinene/caryophyllene oxide group. Interestingly, the 1,8-cineole/camphor group is dominated by samples from Iran. The β-pinene/caryophyllene oxide group, on the other hand, is dominated by samples from Europe. It is apparent that there is much chemical diversity in the essential oils of A. millefolium and that essential oil chemical compositions cannot distinguish New World (i.e., var. occidentalis) samples of A. millefolium from Old World samples.
Figure 2. Dendrogram based on hierarchical cluster analysis of Achillea millefolium essential oil compositions
Kokkalou and co-workers had noted wide variation in A. millefolium essential oil compositions when comparing samples of European origin with samples cultivated in North America [38]. Likewise, Farajpour and co-workers had observed wide variation in A. millefolium essential oil compositions from Iran [58]. These investigators identified five chemotypes: 1,8-cineole/(E)-nerolidol, high cubenol, high germacrene-D/isospathulenol, high camphor/cubenol, and high 1,8-cineole/ β-thujone/cubenol. Furthermore, Raudone and co-workers investigated different morphotypes of A. millefoium and found notable differences in triterpenoid and phenolic components [2].
The A. millefolium essential oils were analyzed by enantioselective GC-MS (Table 3). The dominant enantiomers were (–)-α-pinene (83.2 ± 5.4%), (–)-camphene (98.5 ± 2.2%), (–)-β-pinene (89.5 ± 6.1%), and (–)-α-terpineol (81.8 ± 4.3%). Sabinene, limonene, cis-sabinene hydrate, and trans-sabinene hydrate had variable enantiomeric distributions. The sesquiterpenoids (–)-(E)-β-caryophyllene, (–)-germacrene D, and (+)-(E)-nerolidol were the only enantiomers observed. (–)-Camphor was the only enantiomer observed. When detected, only (–)-β-phellandrene, (–)-α-thujone, (+)-β-thujone, and (–)-borneol were observed. Orth and co-workers carried out enantioselective GC-MS on A. millefolium from Kaliningrad [43]. These investigators found (–)-β-pinene to dominate, but sabinene and α-pinene distributions were variable, although (–)-sabinene and (–)-α-pinene were often dominant.
Table 3. Enantiomeric distribution (percent of each enantiomer) of chiral terpenoid components in Achillea millefolium var. occidentalis.
|
Compounds |
RIdb |
RIcalc |
#1 |
#2 |
#3 |
#4 |
#5 |
#6 |
|
(+)-α-Thujene |
950 |
950 |
nd |
nd |
24.2 |
nd |
0.0 |
nd |
|
(–)-α-Thujene |
951 |
951 |
nd |
nd |
75.8 |
nd |
100.0 |
nd |
|
(–)-α-Pinene |
976 |
977 |
72.8 |
85.7 |
82.0 |
86.0 |
85.4 |
87.3 |
|
(+)-α-Pinene |
982 |
983 |
27.2 |
14.3 |
18.0 |
14.0 |
14.6 |
12.7 |
|
(–)-Camphene |
998 |
1000 |
100.0 |
100.0 |
94.3 |
99.2 |
98.2 |
99.3 |
|
(+)-Camphene |
1005 |
1003 |
0.0 |
0.0 |
5.7 |
0.8 |
1.8 |
0.7 |
|
(+)-Sabinene |
1021 |
1020 |
56.1 |
11.8 |
47.2 |
15.7 |
42.6 |
15.8 |
|
(–)-Sabinene |
1030 |
1029 |
43.9 |
88.2 |
52.8 |
84.3 |
57.4 |
84.2 |
|
(+)-β-Pinene |
1027 |
1027 |
18.8 |
10.5 |
10.6 |
7.7 |
1.1 |
14.6 |
|
(–)-β-Pinene |
1031 |
1031 |
81.2 |
89.5 |
89.4 |
92.3 |
98.9 |
85.4 |
|
(–)-Limonene |
1073 |
1074 |
68.9 |
83.1 |
78.9 |
80.4 |
31.8 |
74.7 |
|
(+)-Limonene |
1081 |
1081 |
31.1 |
16.9 |
21.1 |
19.6 |
68.2 |
25.3 |
|
(–)-β-Phellandrene |
1083 |
1082 |
nd |
nd |
100.0 |
nd |
100.0 |
nd |
|
(+)-β-Phellandrene |
1089 |
nd |
nd |
nd |
0.0 |
nd |
0.0 |
nd |
|
(+)-cis-Sabinene hydrate |
1199 |
1199 |
75.4 |
88.1 |
7.7 |
16.8 |
23.8 |
79.1 |
|
(–)-cis-Sabinene hydrate |
1202 |
1201 |
24.6 |
11.9 |
92.3 |
83.2 |
76.2 |
20.9 |
|
(+)-α-Thujone |
1213 |
nd |
0.0 |
0.0 |
0.0 |
nd |
nd |
nd |
|
(–)-α-Thujone |
1222 |
1221 |
100.0 |
100.0 |
100.0 |
nd |
nd |
nd |
|
(–)-Linalool |
1228 |
1231 |
100.0 |
16.5 |
nd |
nd |
nd |
nd |
|
(+)-Linalool |
1231 |
1234 |
0.0 |
83.5 |
nd |
nd |
nd |
nd |
|
(+)-trans-Sabinene hydrate |
1231 |
1229 |
nd |
nd |
8.1 |
25.9 |
18.5 |
61.5 |
|
(–)-trans-Sabinene hydrate |
1235 |
1233 |
nd |
nd |
91.9 |
74.1 |
81.5 |
38.5 |
|
(+)-β-Thujone |
1230 |
1232 |
100.0 |
nd |
100.0 |
nd |
nd |
nd |
|
(–)-β-Thujone |
na |
nd |
0.0 |
nd |
0.0 |
nd |
nd |
nd |
|
(–)-Camphor |
1253 |
1254 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
|
(+)-Camphor |
1259 |
nd |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
(+)-Terpinen-4-ol |
1297 |
1295 |
60.5 |
59.3 |
26.1 |
21.5 |
31.0 |
61.5 |
|
(–)-Terpinen-4-ol |
1300 |
1297 |
39.5 |
40.7 |
73.9 |
78.5 |
69.0 |
38.5 |
|
(–)-Borneol |
1335 |
1336 |
100.0 |
100.0 |
nd |
nd |
100.0 |
100.0 |
|
(+)-Borneol |
1340 |
nd |
0.0 |
0.0 |
nd |
nd |
0.0 |
0.0 |
|
(–)-α-Terpineol |
1347 |
1346 |
87.6 |
86.1 |
81.7 |
76.9 |
78.1 |
80.7 |
|
(+)-α-Terpineol |
1356 |
1355 |
12.4 |
13.9 |
18.3 |
23.1 |
21.9 |
19.3 |
|
(–)-(E)-β-Caryophyllene |
1461 |
1462 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
|
(+)-(E)-β-Caryophyllene |
na |
nd |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
(+)-Germacrene D |
1519 |
nd |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
(–)-Germacrene D |
1522 |
1523 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
|
(–)-(E)-Nerolidol |
1677 |
nd |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
(+)-(E)-Nerolidol |
1680 |
1679 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
100.0 |
RIdb = Retention index from our in-house database. RIcalc = Calculated retention index based on a homologous series of n-alkanes on a Restek B-Dex 325 capillary column. na = reference compound not available. nd = compound not detected.
There have been several investigations on enantioselective GC-MS analyses of Asteraceae essential oils. However, there are no obvious trends in the enantiomeric distribution of monoterpene hydrocarbons or of oxygenated monoterpenoids in the family [59]. When detected, (–)-(E)-β-caryophyllene is the only enantiomer observed [60–62]. This enantiomer is found in numerous higher plants, while (+)-(E)-β-caryophyllene is apparently found in liverworts [13]. Likewise, (–)-germacrene D and (+)-(E)-nerolidol are the dominant, if not exclusive, enantiomers found in the Asteraceae [59, 61, 62].
Several of the major components in A. millefolium have shown relevant biological activities. For example, 1,8-cineole, α-thujone, β-thujone, and camphor have shown analgesic-like activity [63, 64]. Camphor and 1,8-cineole have shown antitussive effects in a Guinea-pig model [65]. A combination of (–)-camphor and 1,8-cineole has shown synergistic antimicrobial activity against Candida albicans [66]. Terpinen-4-ol has shown antimicrobial activity against several pathogenic micro-organisms [67]. Terpinen-4-ol has also shown anti-inflammatory effects [68], attributed to suppression of pro-inflammatory cytokine production [69, 70]. (E)-Nerolidol has shown antinociceptive activity involving the GABAergic system and not to the opiodergic or ATP-sensitive K+ channels [71]. Nerolidol has also shown anti-inflammatory activity attributed to suppression of TNF-α and IL-1β pro-inflammatory cytokines [71]. Thus, the bioactivities of the major components in A. millefolium essential oil are consistent with the traditional uses of the plant by Native Americans.
4.
Conclusions
The
essential oils of Achillea millefolium var. occidentalis,
collected from several sites in the Great Basin of North America, show wide
variation in their chemical compositions, including broadly variable
concentrations of camphor, α-thujone, terpinen-4-ol, artemisia ketone,
1,8-cineole, and (E)-nerolidol. A survey of the literature also reveals
wide variation in A. millefolium essential oil compositions in samples
from Europe and Asia. Furthermore, because of the wide variation in essential
oil compositions of A. millefolium from the Great Basin as well as the
wide variation in compositions from samples of European or Asian origin,
essential oil compositions cannot distinguish between New-World and Old-World A.
millefolium essential oils. Thus, there is no phytochemical support for
infraspecific separation of this species. Future research using DNA barcoding
may provide evidence for the separation of infraspecific taxa. Enantioselective
gas chromatography has shown (–)-α-pinene, (–)-camphene, (–)-β-pinene,
(–)-α-terpineol, (–)-camphor, (–)-β-phellandrene, (–)-α-thujone, (+)-β-thujone,
and (–)-borneol to be the dominant monoterpenoid enantiomers in A.
millefolium from western North America. However, there are no obvious
trends in the enantiomeric distribution of monoterpenoids in the Asteraceae
family overall. Additional research using enantioselective gas chromatography may
be able to identify trends within individual genera, however.
Authors’ contributions
Conceptualization,
W.N.S.; Methodology, A.P., P.S., and W.N.S.; Software, P.S.; Validation,
W.N.S., Formal Analysis, A.P., and W.N.S.; Investigation, A.P., P.S., K.S., and
W.N.S.; Resources, P.S. and W.N.S.; Data Curation, W.N.S.; Writing – Original
Draft Preparation, W.N.S.; Writing – Review & Editing, P.S. and W.N.S.;
Project Administration, W.N.S.
Acknowledgements
This
work was carried out as part of the activities of the Aromatic Plant Research
Center (APRC, https://aromaticplant.org/).
Funding
This
research received no specific grant from any funding agency.
Availability of data and materials
All
data will be made available on request, according to the journal policy.
Conflicts of interest
The
authors declare no conflict of interest.
References
1.
Bruneton, J. Pharmacognosy; 2nd edition;
Intercept Ltd: London, UK, 1999; ISBN 1-898298-63-7.
2.
Raudone, L.; Vilkickyte, G.; Marksa, M.;
Radusiene, J. Comparative phytoprofiling of Achillea millefolium
morphotypes: Assessing antioxidant activity, phenolic and triterpenic compounds
variation across different plant parts. Plants. 2024, 13, 1043. https://doi.org/10.3390/plants13071043.
3.
World Flora Online, W.F.O. Achillea
millefolium L. Available online:
http://www.worldfloraonline.org/taxon/wfo-0000042097 (accessed on Apr 2, 2023).
4.
Missouri Botanical Garden. Achillea
millefolium. Available online:
https://www.missouribotanicalgarden.org/PlantFinder/PlantFinderDetails.aspx?taxonid=277129&isprofile=1&basic=yarrow
(accessed on Apr 3, 2023).
5.
Winslow, S.R. Western Yarrow, Achillea
millefolium L. var. occidentalis DC.; United States Department of
Agriculture, Natural Resources Conservation Service, Bridger Plant Materials
Center: Bridger, Montana, USA, 2005.
6.
eFloras.org. Flora of North America. Achillea
millefolium Linnaeus. Available online:
http://www.efloras.org/florataxon.aspx?flora_id=1&taxon_id=200023010
(accessed on Apr 2, 2023).
7.
Ali, S.I.; Gopalakrishnan, B.;
Venkatesalu, V. Pharmacognosy, phytochemistry and pharmacological properties of
Achillea millefolium L.: A review. Phyther. Res. 2017,
31, 1140–1161.
https://doi.org/10.1002/ptr.5840.
8.
Train, P.; Henrichs, J.R.; Archer, W.A.
Medicinal Uses of Plants by Indian Tribes of Nevada; U.S. Department of
Agriculture: Washington, DC, USA, 1941.
9.
Fowler, C.S. Willard Z. Park’s
Ethnographic Notes on the Northern Paiute of Western Nevada 1933-1940;
University of Utah Press: Salt Lake City, Utah, USA, 1989.
10.
Bussmann, R.W.; Batsatsashvili, K.;
Kikvidze, Z.; Paniagua-Zambrana, N.Y.; Ghorbani, A.; Nasab, F.K.; Khutsishvili,
M.; Maisaia, I.; Sikharulidze, S.; Tchelidze, D. Achillea grandiflora M.
Bieb. Achillea micrantha Willd. Achillea millefolium L. Achillea
nobilis L. Achillea ptarmicifolia (Willd.) Rupr. ex Heimerl Ptarmica
ptarmicifolia Galushko. In Ethnobotany of the Mountain Regions of Far
Eastern Europe; Batsatsashvili, K., Kikvidze, Z., Bussmann, R.W., Eds.;
Springer Nature: Cham, Switzerland, 2020, pp.47–57. ISBN 978-3-030-28939-3.
11.
Paniagua-Zambrana, N.Y.; Bussmann, R.W.;
Romero, C. Achillea millefolium L. In Ethnobotany of the Andes;
Paniagua-Zambrana, N.Y., Bussmann, R.W., Eds.; Springer Nature: Cham,
Switzerland, 2020, pp.111–115 ISBN 978-3-030-28932-4.
12.
Chandler, R.F.; Hooper, S.N.; Harvey,
M.J. Ethnobotany and phytochemistry of yarrow, Achillea millefolium,
Compositae. Econ. Bot. 1982, 36, 203–223. https://doi.org/10.1007/bf02858720.
13.
Dictionary of Natural Products.
Dictionary of Natural Products on USB; CRC Press: Boca Raton, Florida, 2023;
ISBN 978-0-412-49150-4.
14.
New York Botanical Garden, N.Y.B.G. C.
V. Starr Virtual Herbarium. Available online:
https://sweetgum.nybg.org/science/vh/specimen-list/?SummaryData=Achillea
millefolium (accessed on Jun 30, 2022).
15.
Likens, S.T.; Nickerson, G.B. Detection
of certain hop oil constituents in brewing products. Proceedings. Annu.
Meet. Am. Soc. Brew. Chem. 1964, 22,
5–13. https://doi.org/10.1080/00960845.1964.12006730.
16.
Au-Yeung, C.Y.; MacLeod, A.J. A
comparison of the efficiency of the Likens and Nickerson extractor for aqueous,
lipid/aqueous, and lipid samples. J. Agric. Food Chem. 1981, 29, 502–505. https://doi.org/10.1021/jf00105a016.
17.
Bouseta, A.; Collin, S. Optimized
Likens-Nickerson methodology for quantifying honey flavors. J. Agric. Food Chem.
1995, 43, 1890–1897. https://doi.org/10.1021/jf00055a025.
18.
Satyal, P.; Dosoky, N.S.; Poudel, A.;
Swor, K.; Setzer, W.N. Chemical composition of the aerial parts essential oil
of Chrysothamnus viscidiflorus from southwestern Idaho. J. Essent. Oil
Plant Comp. 2023, 1, 115–121. https://doi.org/10.58985/jeopc.2023.v01i02.16.
19.
Agnihotri, V.K.; Lattoo, S.K.; Thappa,
R.K.; Kaul, P.; Qazi, G.N.; Dhar, A.K.; Saraf, A.; Kapahi, B.K.; Saxena, R.K.;
Agarwal, S.C. Chemical variability in the essential oil components of Achillea
millefolium Agg. from different Himalayan habitats (India). Planta Med.
2005, 71, 280–283. https://doi.org/10.1055/s-2005-837828.
20.
Ahmadi-Dastgerdi, A.; Ezzatpanah, H.;
Asgary, S.; Dokhani, S.; Rahimi, E. Phytochemical, antioxidant and antimicrobial
activity of the essential oil from flowers and leaves of Achillea
millefolium subsp. millefolium. J. Essent. Oil-Bear. Plants. 2017,
20, 395–409. https://doi.org/10.1080/0972060X.2017.1280419.
21.
Ebrahimi, M.; Farajpour, M.; Hadavand,
H.; Bahmani, K.; Khodaiyan, F. Essential oil variation among five Achillea
millefolium ssp. elbursensis collected from different ecological
regions of Iran. Ann. Biol. Res. 2012, 3, 3248–3253.
22.
El-Kalamouni, C.; Venskutonis, P.R.;
Zebib, B.; Merah, O.; Raynaud, C.; Talou, T. Antioxidant and antimicrobial
activities of the essential oil of Achillea millefolium L. grown in
France. Medicines 2017, 4, 30, https://doi.org/10.3390/medicines4020030.
23.
Falconieri, D.; Piras, A.; Porcedda, S.;
Marongiu, B.; Gonçalves, M.J.; Cabral, C.; Cavaleiro, C.; Salgueiro, L.
Chemical composition and biological activity of the volatile extracts of Achillea
millefolium. Nat. Prod. Commun. 2011, 6, 1527–1530. https://doi.org/10.1177/1934578x1100601030.
24.
Farhadi, N.; Babaei, K.; Farsaraei, S.;
Moghaddam, M.; Ghasemi Pirbalouti, A. Changes in essential oil compositions,
total phenol, flavonoids and antioxidant capacity of Achillea millefolium
at different growth stages. Ind. Crops Prod. 2020, 152, 112570. https://doi.org/10.1016/j.indcrop.2020.112570.
25.
Figueiredo, A.C.; Barroso, J.G.; Pais,
M.S.S.; Scheffer, J.J.C. Composition of the essential oils from leaves and
flowers of Achillea millefolium L. ssp. millefolium. Flavour
Fragr. J. 1992, 7, 219–222. https://doi.org/10.1002/ffj.2730070409.
26.
Haziri, A.I.; Aliaga, N.; Ismaili, M.;
Govori-odai, S.; Leci, O.; Faiku, F.; Arapi, V.; Haziri, I. Secondary
metabolites in essential oil of Achillea millefolium (L.) growing wild
in east part of Kosova. Am. J. Biochem. Biotechnol. 2010, 6, 32–34. https://doi.org/10.3844/ajbbsp.2010.32.34.
27.
Howyzeh, M.S.; Aslani, S.; Pooraskari,
O. Essential oil profile of an Iranian yarrow (Achillea millefolium). J.
Essent. Oil-Bear. Plants. 2019, 22, 295–300. https://doi.org/10.1080/0972060X.2019.1586589.
28.
Jaimand, K.; Rezaee, M.B.; Mozaffarian,
V. Chemical constituents of the leaf and flower oils from Achillea
millefolium ssp. elbursensis Hub.-Mor. from Iran rich in
chamazulene. J. Essent. Oil Res. 2006, 18, 293–295, https://doi.org/10.1080/10412905.2006.9699093.
29.
Judzentiene, A.; Mockute, D. Essential
oil composition of two yarrow taxonomic forms. Cent. Eur. J. Biol. 2010, 5,
346–352. https://doi.org/10.2478/s11535-010-0011-7.
30.
Kokkalou, E.; Kokkini, S.; Hanlidou, E.
Volatile constituents of Achillea millefolium in relation to their
infraspecific variation. Biochem. Syst. Ecol. 1992, 20, 665–670,
doi:10.1016/0305-1978(92)90023-7.
31.
Azizi, M.; Chizzola, R.; Ghani, A.;
Oroojalian, F. Composition at different development stages of the essential oil
of four Achillea species grown in Iran. Nat. Prod. Commun. 2010, 5,
283–290, https://doi.org/10.1177/1934578x1000500224.
32.
Konarska, A.; Weryszko-Chmielewska, E.;
Sulborska-Różycka, A.; Kiełtkya-Dadasiewicz, A.; Dmitruk, M.; Gorzel, M. Herb
and flowers of Achillea millefolium subsp. millefolium L.:
Structure and histochemistry of secretory tissues and phytochemistry of essential
oils. Molecules. 2023, 28, 7791. https://doi.org/10.3390/molecules28237791.
33.
Nadim, M.M.; Malik, A.A.; Ahmad, J.;
Bakshi, S.K. The essential oil composition of Achillea millefolium L.
cultivated under tropical condition in India. World J. Agric. Sci. 2011, 7,
561–565.
34.
Nenaah, G.E. Chemical composition,
toxicity and growth inhibitory activities of essential oils of three Achillea
species and their nano-emulsions against Tribolium castaneum (Herbst).
Ind. Crops Prod. 2014, 53, 252–260, https://doi.org/10.1016/j.indcrop.2013.12.042.
35.
Orav, A.; Kailas, T.; Ivask, K.
Composition of the essential oil from Achillea millefolium L. from
Estonia. J. Essent. Oil Res. 2001, 13, 290–294. https://doi.org/10.1080/10412905.2001.9699697.
36.
Orth, M.; Czygan, F.C.; Dedkov, V.P.
Variation in essential oil composition and chiral monoterpenes of Achillea
millefolium s.l. from Kaliningrad. J. Essent. Oil Res. 1999, 11, 681–687. https://doi.org/10.1080/10412905.1999.9711995.
37.
Pino, J.A.; Rosado, A.; Fuentes, V.
Chemical composition of the leaf oil of Achillea millefolium L. grown in
Cuba. J. Essent. Oil Res. 1998, 10, 427–428. https://doi.org/10.1080/10412905.1998.9700934.
38.
Rahimmalek, M.; Tabatabaei, B.E.S.;
Etemadi, N.; Goli, S.A.H.; Arzani, A.; Zeinali, H. Essential oil variation among
and within six Achillea species transferred from different ecological
regions in Iran to the field conditions. Ind. Crops Prod. 2009, 29, 348–355. https://doi.org/10.1016/j.indcrop.2008.07.001.
39.
Rowshan, V.; Bahmanzadegan, A. Effects
of salicylic acid on essential oil components in yarrow (Achillea
millefolium Boiss). Int. J. Basic Sci. Appl. Res. 2013, 2, 347–351.
40.
Shawl, A.S.; Srivastava, S.K.;
Syamasundar, K. V.; Tripathi, S.; Raina, V.K. Essential oil composition of Achillea
millefolium L. growing wild in Kashmir, India. Flavour Fragr. J. 2002, 17,
165–168. https://doi.org/10.1002/ffj.1074.
41.
Smelcerovic, A.; Lamshoeft, M.;
Radulovic, N.; Ilic, D.; Palic, R. LC-MS analysis of the essential oils of Achillea
millefolium and Achillea crithmifolia. Chromatographia. 2010, 71,
113–116. 10.1365/s10337-009-1393-4.
42.
Ben Abdallah, S.; Riahi, C.; Vacas, S.;
Navarro-Llopis, V.; Urbaneja, A.; Pérez-Hedo, M. The dual benefit of plant
essential oils against Tuta absoluta. Plants. 2023, 12, 985. https://doi.org/10.3390/plants12050985.
43.
Stevanovic, Z.D.; Pljevljakušic, D.;
Ristic, M.; Šoštaric, I.; Kresovic, M.; Simic, I.; Vrbnièanin, S. Essential oil
composition of Achillea millefolium agg. populations collected from
saline habitats in Serbia. J. Essent. Oil-Bear. Plants. 2015, 18, 1343–1352. https://doi.org/10.1080/0972060X.2014.884952.
44.
Suleimenov, Y.M.; Atazhanova, G.A.;
Ozek, T.; Demirci, B.; Kulyyasov, A.T.; Adekenov, S.M.; Baser, K.H.C. Essential
oil composition of three species of Achillea from Kazakhstan. Chem. Nat.
Compd. 2001, 37, 447–450.
45.
Verma, R.S.; Joshi, N.; Padalia, R.C.;
Goswami, P.; Singh, V.R.; Chauhan, A.; Verma, S.K.; Iqbal, H.; Verma, R.K.;
Chanda, D.; Sundaresan, V.; Darokar, M.P. Chemical composition and
allelopathic, antibacterial, antifungal and in vitro acetylcholinesterase
inhibitory activities of yarrow (Achillea millefolium L.) native to
India. Ind. Crops Prod. 2017, 104, 144–155. https://doi.org/10.1016/j.indcrop.2017.04.046.
46.
Mazandarani, M.; Mirdeilami, S.Z.;
Pessarakli, M. Essential oil composition and antibacterial activity of Achillea
millefolium L. from different regions in North east of Iran. J. Med. Plants
Res. 2013, 7, 1063–1069. https://doi.org/10.5897/JMPR12.961.
47.
Bertoli, A.; Conti, B.; Mazzoni, V.;
Meini, L.; Pistelli, L. Volatile chemical composition and bioactivity of six
essential oils against the stored food insect Sitophilus zeamais Motsch.
(Coleoptera Dryophthoridae). Nat. Prod. Res. 2012, 26, 2063–2071. https://doi.org/10.1080/14786419.2011.607453.
48.
Boskovic, Z.; Radulovic, N.; Stojanovic,
G. Essential oil composition of four Achillea species from the Balkans
and its chemotaxonomic significance. Chem. Nat. Compd. 2005, 41, 674–678. https://doi.org/10.1007/s10600-006-0009-6.
49.
Candan, F.; Unlu, M.; Tepe, B.;
Daferera, D.; Polissiou, M.; Sökmen, A.; Akpulat, H.A. Antioxidant and
antimicrobial activity of the essential oil and methanol extracts of Achillea
millefolium subsp. millefolium Afan. (Asteraceae). J.
Ethnopharmacol. 2003, 87, 215–220. https://doi.org/10.1016/S0378-8741(03)00149-1.
50.
Czerniewicz, P.; Chrzanowski, G.;
Sprawka, I.; Sytykiewicz, H. Aphicidal activity of selected Asteraceae
essential oils and their effect on enzyme activities of the green peach aphid, Myzus
persicae (Sulzer). Pestic. Biochem. Physiol. 2018, 145, 84–92. https://doi.org/10.1016/j.pestbp.2018.01.010.
51.
Daniel, P.S.; Lourenço, E.L.B.; da Cruz,
R.M.S.; De Souza Gonçalves, C.H.; Das Almas, L.R.M.; Hoscheid, J.; da Silva,
C.; Jacomassi, E.; Brum Junior, L.; Alberton, O. Composition and antimicrobial
activity of essential oil of yarrow (Achillea millefolium L.). Aust. J.
Crop Sci. 2020, 14, 545–550. https://doi.org/10.21475/ajcs.20.14.03.p2325.
52.
de Sant’Anna, J.R.; da Silva Franco,
C.C.; Miyamoto, C.T.; Cunico, M.M.; Miguel, O.G.; Côcco, L.C.; Yamamoto, C.I.;
Corrêa Junior, C.; de Castro-Prado, M.A.A. Genotoxicity of Achillea
millefolium essential oil in diploid cells of Aspergillus nidulans.
Phyther. Res. 2008, 22, 544–549. https://doi.org/10.1002/ptr.2596.
53.
van den Dool, H.; Kratz, P.D. A
generalization of the retention index system including linear temperature
programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11,
463–471.
https://doi.org/10.1016/S0021-9673(01)80947-X.
54.
Adams, R.P. Identification of Essential
Oil Components by Gas Chromatography/Mass Spectrometry; 4th ed.; Allured
Publishing: Carol Stream, IL, USA, 2007; ISBN 978-1-932633-21-4.
55.
Mondello, L. FFNSC 3; Shimadzu
Scientific Instruments: Columbia, Maryland, USA, 2016.
56.
NIST20; National Institute of Standards
and Technology: Gaithersburg, Maryland, USA, 2020.
57.
Satyal, P. Development of GC-MS Database
of Essential Oil Components by the Analysis of Natural Essential Oils and
Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in
Essential Oils, Ph.D. dissertation, University of Alabama in Huntsville,
Huntsville, AL, USA, 2015.
58.
Farajpour, M.; Ebrahimi, M.; Baghizadeh,
A.; Aalifar, M. Phytochemical and yield variation among Iranian Achillea
millefolium accessions. Hort. Sci. 2017, 52, 827–830. https://doi.org/10.21273/HORTSCI11654-16.
59.
Poudel, A.; Satyal, P.; Swor, K.;
Setzer, W.N. Essential oils of two Great Basin composites: Chaenactis
douglasii and Dieteria canescens from southwestern Idaho. J. Essent.
Oil Plant Comp. 2023, 1, 275–283. https://doi.org/10.58985/jeopc.2023.v01i03.35.
60.
Swor, K.; Satyal, P.; Poudel, A.;
Setzer, W.N. Chemical characterization of three Artemisia tridentata
essential oils and multivariate analyses: A preliminary investigation. Nat.
Prod. Commun. 2023, 18, 1934578X231154965. https://doi.org/10.1177/1934578X231154965.
61.
Swor, K.; Satyal, P.; Poudel, A.;
Setzer, W.N. The essential oil of Balsamorhiza sagittata from
southwestern Idaho: Chemical composition and enantiomeric distribution. Nat.
Prod. Commun. 2024, 19, 1934578X231225842. https://doi.org/10.1177/1934578X231225842.
62.
Swor, K.; Satyal, P.; Poudel, A.;
Setzer, W.N. The essential oils of Wyethia species: Wyethia
amplexicaulis (Nutt.) Nutt. and Wyethia helianthoides Nutt. Nat.
Prod. Commun. 2024, 19, 1934578X241248081. https://doi.org/10.1177/1934578X241248081.
63.
de Sousa, D.P. Analgesic-like activity
of essential oils constituents. Molecules 2011, 16, 2233–2252. https://doi.org/10.3390/molecules16032233.
64.
Craven, R. The comfort of camphor. Nat.
Rev. Neurosci. 2005, 6, 826. https://doi.org/10.1038/nrn1801.
65.
Laude, E.A.; Morice, A.H.; Grattan, T.J.
The antitussive effects of menthol, camphor and cineole in conscious
Guinea-pigs. Pulm. Pharmacol. 1994, 7, 179–184. https://doi.org/10.1006/pulp.1994.1021.
66.
Viljoen, A.; Van Vuuren, S.; Ernst, E.;
Klepser, M.; Demirci, B.; Başer, H.; Van Wyk, B.E. Osmitopsis asteriscoides
(Asteraceae)-the antimicrobial activity and essential oil composition of a
Cape-Dutch remedy. J. Ethnopharmacol. 2003, 88, 137–143. https://doi.org/10.1016/S0378-8741(03)00191-0.
67.
Johansen, B.; Duval, R.E.; Sergere, J.C.
First evidence of a combination of terpinen-4-ol and α-terpineol as a promising
tool against ESKAPE pathogens. Molecules. 2022, 27, 7472. https://doi.org/10.3390/molecules27217472.
68.
Pongprayoon, U.; Soontornsaratune, P.;
Jarikasem, S.; Sematong, T.; Wasuwat, S.; Claeson, P. Topical antiinflammatory
activity of the major lipophilic constituents of the rhizome of Zingiber
cassumunar. Part I: The essential oil. Phytomed. 1997, 3, 319.322. https://doi.org/10.1016/S0944-7113(97)80003-7.
69.
Hart, P.H.; Brand, C.; Carson, C.F.;
Riley, T. V.; Prager, R.H.; Finlay-Jones, J.J. Terpinen-4-ol, the main
component of the essential oil of Melaleuca alternifolia (tea tree oil),
suppresses inflammatory mediator production by activated human monocytes.
Inflamm. Res. 2000, 49, 619–626. https://doi.org/10.1007/s000110050639.
70. Nogueira, M.N.M.; Aquino, S.G.; Rossa, C.; Spolidorio, D.M.P. Terpinen-4-ol and alpha-terpineol (tea tree oil components) inhibit the production of IL-1β, IL-6 and IL-10 on human macrophages. Inflamm. Res. 2014, 63, 769–778. https://doi.org/10.1007/s00011-014-0749-x.
71. Fonsêca, D. V.; Salgado, P.R.R.; de Carvalho, F.L.; Salvadori, M.G.S.S.; Penha, A.R.S.; Leite, F.C.; Borges, C.J.S.; Piuvezam, M.R.; Pordeus, L.C. de M.; Sousa, D.P.; Almeida, R.N.. Nerolidol exhibits antinociceptive and anti-inflammatory activity: Involvement of the GABAergic system and proinflammatory cytokines. Fundam. Clin. Pharmacol. 2016, 30, 14–22. https://doi.org/10.1111/fcp.12166.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Achillea
millefolium is native to temperate regions of the
northern hemisphere and is important in traditional herbal medicines throughout
its range. The purpose of this work is to evaluate the essential oil
composition of the North American variety, A. millefolium var. occidentalis,
and to test the hypothesis that var. occidentalis has a chemical
composition different from Old World A. millefolium. Six samples of A.
millefolium were collected from different locations in the Great Basin
region of eastern Oregon and southern Idaho. The essential oils were obtained
by hydrodistillation and analyzed by gas chromatographic methods. The essential
oils showed wide variation in composition with the major components (–)-camphor
(0.5-59.6%), (–)-α-thujone (0.0-40.5%), terpinen-4-ol (0.9-23.4%), artemisia
ketone (0.0-18.7%), 1,8-cineole (5.3-16.4%), and (+)-(E)-nerolidol
(0.4-13.5%). The essential oil compositions of western North American A.
millefolium cannot be distinguished from the samples from Old World samples
of A. millefolium; there is no phytochemical support for infraspecific
differentiation of the North American variety.
Abstract Keywords
Yarrow,
chemotaxonomy, gas chromatography, mass spectrometry, enantiomers.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


