Research Article
Paul Abuh Omale
Paul Abuh Omale
1.
Department of Agricultural and
Environmental Engineering, Federal University of Agriculture Makurdi, Benue
State, Nigeria.
2.
Department of Agricultural and
Environmental Engineering, University of Ibadan, Oyo State, Nigeria.
3.
Department of Biosystems and
Agricultural Engineering, University of Kentucky, Lexington, USA
E-mail:
Omale.paul@uam.edu.ng.
Ademola Kabir Aremu
Ademola Kabir Aremu
Department of
Agricultural and Environmental Engineering, University of Ibadan, Oyo State,
Nigeria.
E-mail:
ademolaomooroye@gmail.com
Mobolaji Oluyimika Omobowale
Mobolaji Oluyimika Omobowale
Department of Agricultural
and Environmental Engineering, University of Ibadan, Oyo State, Nigeria.
E-mail:
yimikaomo@gmail.com
Tosin Olayemi Olanrewaju
Tosin Olayemi Olanrewaju
Department of
Biosystems and Agricultural Engineering, University of Kentucky, Lexington, USA
E-mail: tosin.Olanrewaju@uky.edu
Akinbode A Adedeji
Akinbode A Adedeji
Corresponding
Author
Department of
Biosystems and Agricultural Engineering, University of Kentucky, Lexington, KY.
40546, USA
E-mail: akinbode.adedeji@uky.edu;
Tel: +1-859-218-4355
Abstract
Tiger nuts (Cyperus esculentus) are underutilized edible oil seeds
cultivated across Africa with potential as a source of healthy vegetable oil.
It is rich in oil with about 22.14 – 44.92% of the dry weight. This study’s main
aim was to evaluate the quality of tiger nut oil and determine the best variety
and processing conditions for producing high-quality from it to meet the
increasing demand for healthier and more sustainable vegetable oils. Applying a completely randomized
design, oil was extracted
mechanically using an oil expeller and the impact of variety (yellow and brown), moisture level (7%, 10% and 13%), toasting temperature (50 °C, 70 °C and 90 °C) and toasting time (10, 20 and 30
min) were investigated. Using standard procedures, five oil quality parameters
were determined, namely free fatty acid (FFA),
iodine value (IV), peroxide value (PV), refractive index (RI) and
relative density (RD). The result shows a range of 0.22 - 0.49% for FFA, 52.62 – 80.20 (mg/100g) for IV, 2.18-
4.52 (meqO2/Kg) for PV, 1.45 - 1.47 for RI,
and 0.91 - 0.98 for RD. FFA and PV
all increased as the moisture level, toasting time and toasting temperature increased,
whereas the IV decreases. The RI and RD values
decreased as the toasting temperature and toasting time increased but
increased as the moisture level increased. ANOVA
indicates that variety, moisture
level, toasting temperature and toasting time have
significant effects on the FFA, IV,
PV, RI and RD at P ≤0.05. The results show that the best oil quality was achieved at 7 % moisture content, 50 °C
toasting temperature and 10 min toasting time for both varieties. The
brown tiger nut has more quality oil than the yellow Tiger nut. The tiger nut oil has comparable characteristics
common to vegetable oils used in food related
applications and can be considered a viable
alternative to common vegetable oils for food preparation. Findings from this study will help tiger nut
oil processors to make the right processing pretreatment before oil extraction.
Abstract Keywords
Free fatty acid, iodine value, peroxide value,
tiger nut, tiger nut oil
1. Introduction
Tiger-nut (Cyperus esculentus L.) is a species of
plant of the Cyperaceae family and is a native to Egypt cultivated for its
nutritional and health value [1,
2]. Tiger nut according to
regional variation, historical influence and indigenous languages is called
"chufa", "yellow nutsedge", "earth almond" and
"ground almond" in other nations [3], while
in Nigeria, it’s named based on different ethnicity as “Shoho” by the Tiv
ethnic group, “Aya” by the Hausa, “Ofio” by the Yoruba and “Akiausa” by the
Igbo [1]. Globally,
about 9000 metric tons of tiger-nut are produced annually. It has three
varieties namely, black, brown, and yellow [4]. In most cultures, it is consumed as is
(snack) and more recently in the production of non-dairy milk. There is growing
interest in its potential health benefits and applications in different food
applications. Tiger nut is a good source of antioxidants that protects the body
against ageing and ailments such as cancer and heart diseases, protein,
unsaturated oil, starch, fiber, essential minerals such as magnesium and
potassium, vitamin C and E among others. These made it highly important in the
food and medical industries as reported by [5] but
in Africa especially Nigeria, tiger nut
is underutilized and only processed locally into milk or consumed fresh or
dried, whereas it can be processed into other forms such as oil, cake,
biscuits, bread, and flour.
Fats and oil are
important components of both human food and animal feed that serves as nutrient
needed by the human body which are obtainable from oil seeds and nuts with
different oil compositions [6, 7]. Tiger nut has 22.14 - 44.92 % of oil by
dry weight as reported by Makareviciene et al. [8] and it is rich in essential fatty acids
like oleic and linoleic acid, tocopherols (Vitamin E), free of Gluten, and a
good moisturizer. During storage, it is reported that tiger nut starch content
decreases while the reducing sugar content increases [9]. Tiger-nut oil is said to be very stable
under storage without direct light and has a golden brown or golden yellow
coloration depending on the variety of Tiger nut it was obtained from. The oil
has nutty taste, sweet aroma and contain about 18 % saturated and 82 %
unsaturated fatty acids [10], which
makes a healthier oil option. Ezebor et al. [11] reported
that tiger nut oil has high content of oleic acid and low polyunsaturated fatty
acid, low acidity, and a lot of monounsaturated fatty acids. Aremu et al. [6] reported that the demand for edible oil is on
the increase in Africa, and they rely majorly on vegetable oils such as palm
oil, soya bean oil, and groundnut oil among others for meeting this need. Tiger
nut oil could be another option for consumers if its oil quality properties are
well defined [12]. Tiger nut being an ancient plant has not been studied
exhaustively in ways that other similar plant oils have been studied. There are
some research done to evaluate its physicochemical properties, and novel uses [13], but there is a need to understand and
evaluate the effects of varieties and processing conditions on tiger nut oil
quality and stability. Therefore, the objective of this study
was to investigate tiger nut oil quality
attributes mechanically extracted based on its varieties (brown and yellow) and
determine how select processing conditions namely, moisture level, toasting
temperature and toasting time, affect these quality attributes.
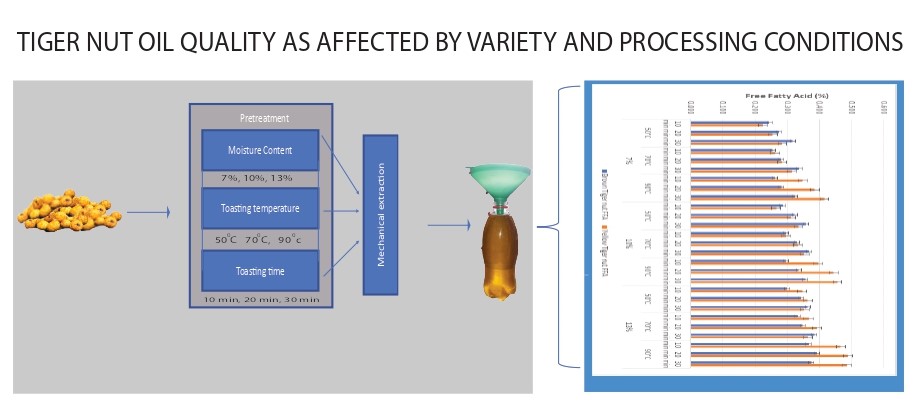
2. Materials and methods
2.1 Sample acquisition and preparations
Yellow
and brown tiger nuts were purchased directly from a tiger nut farm in Katsina
State, Northern part of Nigeria, and were cleaned of all dirts and foreign
matters. The initial
moisture content was determined following the [14] procedure
of drying at 135 °C for 2 h. A total of one hundred and sixty two (162) runs of
2 kg each were applied (324 kg sample size). Eighty one (81) runs of 2 kg each
of the brown and yellow varieties of tiger nut were conditioned (by
rehydration) into three desired moisture levels of 7%, 10% and 13% (db) each of
twenty seven (27) runs applying equation 1 as used by [14]
and [15].
1
Where,
Q is the mass of
moisture to be added in g,
W1 is the initial mass of the sample in g,
Mi is the
initial moisture content of the sample and
Mf is the desired (final) moisture content
of the sample.
Each treatment in three (3)
replications were heated (toasted) at 50 °C, 70 °C and 90 °C toasting
temperature for 10 min, 20 min and 30 min toasting time respectively before the
oil extraction was performed.
2.2 Determination
of oil quality properties
Oil
was extracted from the various prepared samples using an electrically powered
motorized tiger nut oil expeller with temperature regulated barrel developed at
the University of Ibadan-Nigeria. The oil was analyzed for free fatty acid
(FFA), iodine value (IV), peroxide value (PV), refractive index (RI) and
Relative density following standard procedures in order to investigate the
effect of variety, moisture level and toasting time on the oil quality. These analyses
were done at the Food Engineering Laboratory of the Department of Biosystems
and Agricultural Engineering, University of Kentucky, Lexington, USA because of
the interest in quality assurance, access to advanced analytical techniques and
collaboration with experts. All reagents and solvents used for this study were
supplied by Fisher Scientific and VWR in USA to the above stated lab.
2.2.1 Determination
of free fatty acids (FFA) content
This was achieved using the AOAC Official Method 940.28 [17] and the procedure reported by Ogori et al. [18]. Two grams of the tiger nut oil sample was
mixed with 50 mL of 95 % neutral ethyl alcohol
and swirled. One to two milliliters of Phenolphthalein
were added as an indicator. The solution
was titrated with 0.1 N sodium hydroxide until pinkish color
was observed and it terminates. The
volume (V) of NaOH required to produce the first permanent pink color was
recorded to evaluate the free fatty acid (FFA) content of the oil applying
equation 2.
2
V
= volume of NaOH used (mL),
N
= Normality (concentration) of NaOH (0.1 N),
w
= weight of oil used (g)
2.2.2 Determination
of iodine value
Iodine
value of the extracted tiger nut oil was determined following the method
described by AOAC Official Method 941.21 [19] and ISO 3961:2018 [20]. About 2 g of the oil was
delivered to a 300 mL conical flask with ground-in stopper and was mixed with
25.0 mL carbon tetrachloride and sealed.
25.0 mL Hanus solution was added and sealed, it was shaken for one
minute and left in a dark room for 30 minutes with occasional shaking. 10.0 mL of 15% potassium iodide and 100 mL
water (boiled and cooled) were added, sealed, and shaken for 30 seconds. 1 mL of soluble starch was added
and the mixture titrated with 0.1 mol/l
sodium thiosulfate to obtain iodine value when the blue color changed to milky
white or colorless. Likewise, a blank test was performed to obtain
blank level. The iodine value was
calculated using Equation 3.
3
Where
a =Volume (mL) of 0.1 mol/l sodium
thiosulfate consumed in the blank
test, b = Volume (mL) of 0.1 mol/l sodium thiosulfate
consumed in the test, N = Normality of sodium thiosulfate,
W = Weight of sample (g),
2.2.3 Determination
of peroxide
The
Peroxide values (PV) of tiger nut oil was determined
according to ISO 3960:2017
[21, 18]. The
oil sample (5 g) was
weighed into a 200 mL conical flask and
mixed with 30 mL of glacial acetic acid and 20 mL of chloroform and mixed
thoroughly by swirling the flask.
Potassium iodide (0.5 mL) was added,
and the mixture was
left in the dark for 1 minute with
occasional swirling, followed with further addition of 30 mL of distilled water. The mixture was titrated
with 0.1 N sodium thiosulfate solution with 0.5 mL of 1.0% soluble starch as
indicator until the dark blue color disappears.
A blank sample titration was carried
out in the same manner but with no oil added.
The Peroxide
values (PV) were
calculated using Equation 4 below.
4
Where,
a =Volume (mL) of 0.1 mol/l sodium
thiosulfate consumed in the blank
test, b = Volume
(mL)
of 0.1 mol/l sodium
thiosulfate consumed in the test, N
= Normality of sodium thiosulfate and W = Weight of sample.
2.2.4 Determination
refractive index
N-1E Handheld Refractometer (Atago, Japan) was used in this
determination as described by ISO 6320:2017 [22]. Before
introducing the sample, water at 30 °C was circulated around the glass slide to
keep its temperature uniform and through the eyepiece of the refractometer, the
dark portion viewed was adjusted to be in line with the intersection of the
cross. One drop of the tiger nut oil sample was transferred into the glass
slide (prism) of the refractometer after proper cleaning with ethanol and
cotton. The sample is allowed to spread all over the prism surface, the scale
was read where the boundary line intercepts the scale by looking through the
eyepiece and recorded as the refractive index of the sample and the mean values
calculated.
2.2.5 Determination
of relative density
The
procedures for specific density determined followed by [23] was applied. Density
bottle was used to determine the density of the tiger nut oil. A clean and dry
bottle of 25 mL capacity was weighed (W0) and then filled with the
oil, stopper inserted and reweighed to give (W1). The oil was
substituted with water after washing and drying the bottle and weighed to give
(W2). The specific gravity (Sp.gr) was calculated applying equation
5.
5
Where W0 = Weight of container, g, W1 = Weight of container and oil, g, W2 = Weight of container and water, g,
2.3 Oil
quality data analysis
The oil quality properties were determined in four
replications and statistically analyzed at 95% confidence level using SPSS
version 20 software (IBM, USA) and a Duncan test was performed to separate the
means to identify the major areas of significance.
3. Results and
discussion
3.1 Effects of
variety, moisture level and toasting time on Free Fatty Acid
The free fatty acid (FFA) from this study ranged from
0.22-0.49% as presented in Table 1, with the tiger nuts at 7% moisture level
toasted for 10 min at toasting temperature of 50°C
having the least values, while tiger nut at 13% toasted for 30 min at 90°C
toasting temperature had the highest FFA values. The results show that the FFA
increases as the moisture level, toasting time and toasting temperature increases.
Analysis of variance (ANOVA) revealed that variety, moisture level, toasting
time and toasting temperature all have significant effects on the FFA value at p<0.05.
The range of FFA values obtained in this study are all <0.6 % which is the
recommended range of FFA for fats and oil by the World Health Organization and
Food and Agricultural Organization of the United Nations. [18] reported a range of 0.39 - 0.41(%) and 0.19 -
0.22 (%) for stored brown and yellow tiger nut oil and this report is slightly
low from the result observed in this study. This difference could be because of
the different oil extraction methods used and the effect of the heating
temperature and time applied in this study.
The reports are similar in line with the report by [24] for tiger nut, [25]
report for sesame seed oil, report on roasted soybean by [26]. The low FFA values obtained in this study are
an indication that tiger nut oil would be stable over long period of time without
rancidity and peroxidation as reported by [6] for
several oil seeds in Nigeria.
Table 1. Tiger nut oil free fatty acid (FFA)
investigated as influenced by variety, moisture level, toasting temperature and
toasting time.
|
Moisture
Level (%) |
Toasting
Temperature (°C) |
Toasting Time (min) |
Brown Tiger nut FFA
(%) |
Standard Deviation |
Yellow Tiger nut
FFA |
Standard Deviation |
|
7 |
50 |
10 |
0.24aaa |
0.004 |
0.22aaa |
0.003 |
|
20 |
0.27aab |
0.001 |
0.25aab |
0.001 |
||
|
30 |
0.32aac |
0.002 |
0.28aac |
0.002 |
||
|
70 |
10 |
0.25aba |
0.004 |
0.26aba |
0.001 |
|
|
20 |
0.28abb |
0.001 |
0.28abb |
0.001 |
||
|
30 |
0.34abc |
0.002 |
0.31abc |
0.002 |
||
|
90 |
10 |
0.26aca |
0.001 |
0.35aca |
0.003 |
|
|
20 |
0.28acb |
0.001 |
0.39acb |
0.003 |
||
|
30 |
0.32acc |
0.002 |
0.41acc |
0.001 |
||
|
10 |
50 |
10 |
0.29baa |
0.004 |
0.27baa |
0.017 |
|
20 |
0.32bab |
0.000 |
0.31bab |
0.004 |
||
|
30 |
0.36bac |
0.001 |
0.33bac |
0.003 |
||
|
70 |
10 |
0.30bba |
0.004 |
0.30bba |
0.001 |
|
|
20 |
0.33bbb |
0.003 |
0.33bbb |
0.002 |
||
|
30 |
0.37bbc |
0.001 |
0.35bbc |
0.001 |
||
|
90 |
10 |
0.30bca |
0.001 |
0.40bca |
0.001 |
|
|
20 |
0.33bcb |
0.001 |
0.44bcb |
0.008 |
||
|
30 |
0.36bcc |
0.001 |
0.46bcc |
0.002 |
||
|
13 |
50 |
10 |
0.30caa |
0.022 |
0.35caa |
0.003 |
|
20 |
0.34cab |
0.001 |
0.36cab |
0.000 |
||
|
30 |
0.36cac |
0.003 |
0.35cac |
0.002 |
||
|
70 |
10 |
0.33cba |
0.001 |
0.37cba |
0.002 |
|
|
20 |
0.35cbb |
0.001 |
0.39cbb |
0.003 |
||
|
30 |
0.38cbc |
0.002 |
0.36cbc |
0.002 |
||
|
90 |
10 |
0.37cca |
0.002 |
0.47cca |
0.002 |
|
|
20 |
0.39ccb |
0.003 |
0.49ccb |
0.009 |
||
|
30 |
0.37ccc |
0.002 |
0.49ccc |
0.020 |
||
|
Note; The first, second and third letters
represents the effects of moisture content, toasting temperature and toasting
time on oil quality respectively.
Means with the same superscript are not significantly different along
the same column. |
||||||
3.2 Effects
of variety, moisture level and toasting time on Iodine value
The experimental result of Iodine value is reported in Table
2, and it ranged from 52.62-80.20(mg/100 g) with the brown tiger nut at 13%
moisture level toasted for 30 min at 70°C toasting temperature having the
highest value of iodine while the least value was obtained from the yellow
tiger nut at 7% moisture level toasted for 10 min at 90°C toasting temperature.
The moisture level, toasting time, toasting temperature, and variety were
reported to have significant effect on the iodine value at p<0.05
after Analysis of variance. The range of iodine value obtained in this research
is within the acceptable iodine value for olive oil published by [27] fats and oil regulations and [28] for existing named vegetable oils. The iodine
value of tiger nut oil from this study is below 100 mg/100g and it indicates
that the tiger nut oil is a nondrying oil (it does not harden but remains in
liquid form when exposed to air) [6]. [29] reported
67.35 mg/100g as iodine value of tiger nut oil extracted mechanically and this
value is within the value range reported in this study. [30, 31] reported
similar observations in their study of rapeseed oil and canola oil
respectively. The differences in the results may be as a result of the
treatment the tiger nut was exposed to before extraction of its oil. This
result is in line with the iodine value of Castor seed oil reported by [32].
Table 2. Iodine value (IV) of tiger nut oil investigated as influenced by variety, moisture level,
toasting temperature and toasting time.
|
Moisture Level (%) |
Toasting Temp (°C) |
Toasting Time (min) |
BT nut IV (mg/100g) |
Standard Deviation |
YT nut IV (mg/100g) |
Standard Deviation |
|
7 |
50 |
10 |
76.83aca |
0.045 |
76.46aca |
0.044 |
|
20 |
76.33acb |
0.024 |
76.27acb |
0.040 |
||
|
30 |
76.17acc |
0.035 |
76.19acc |
0.009 |
||
|
70 |
10 |
66.83aba |
0.045 |
56.62aba |
0.325 |
|
|
20 |
70.33abb |
0.024 |
60.35abb |
0.559 |
||
|
30 |
75.21abc |
0.045 |
70.27abc |
0.090 |
||
|
90 |
10 |
57.63aaa |
0.315 |
52.62aaa |
0.325 |
|
|
20 |
61.36aab |
0.556 |
55.69aab |
0.165 |
||
|
30 |
71.27aac |
0.094 |
60.27aac |
0.090 |
||
|
10 |
50 |
10 |
78.47bca |
0.014 |
78.74bca |
0.040 |
|
20 |
78.14bcb |
0.031 |
77.84bcb |
0.003 |
||
|
30 |
77.45bcc |
0.017 |
77.24bcc |
0.026 |
||
|
70 |
10 |
74.47bba |
0.010 |
62.27bba |
0.029 |
|
|
20 |
77.13bbb |
0.016 |
67.89bbb |
0.609 |
||
|
30 |
78.23bbc |
0.062 |
74.85bbc |
0.467 |
||
|
90 |
10 |
63.28baa |
0.037 |
58.27baa |
0.029 |
|
|
20 |
68.77bab |
0.636 |
64.55bab |
0.357 |
||
|
30 |
75.84bac |
0.480 |
68.85bac |
0.666 |
||
|
13 |
50 |
10 |
79.84cca |
0.061 |
79.85cca |
0.004 |
|
20 |
79.54ccb |
0.031 |
79.21ccb |
0.014 |
||
|
30 |
79.13ccc |
0.036 |
78.88ccc |
0.029 |
||
|
70 |
10 |
78.17cba |
0.539 |
68.45cba |
0.022 |
|
|
20 |
79.16cbb |
0.040 |
72.32cbb |
0.050 |
||
|
30 |
80.20cbc |
0.622 |
77.32cbc |
0.592 |
||
|
90 |
10 |
69.46caa |
0.014 |
65.45caa |
0.022 |
|
|
20 |
73.32cab |
0.050 |
70.15cab |
0.279 |
||
|
30 |
78.32cac |
0.586 |
75.66cac |
0.020 |
||
|
Note; The first, second and third letters
in the superscript represents the effects of moisture content, toasting
temperature and toasting time on oil quality respectively. Means with the same superscript are not
significantly different along the same column. BT – Brown Tiger; YT – Yellow
Tiger; Temp – Temperature; |
||||||
3.3 Effects of
variety, moisture level and toasting time on Peroxide value
Peroxide value is an important quality parameter that
deduces oxidation of the oil [33]. The
peroxide value of the experimental samples analyzed ranged from 2.18-4.52 (meqO2/Kg)
as shown in Table 3, with the tiger nuts having their least peroxide values at
13% moisture level toasted for 10 min at toasting temperature of 50°C and the
highest peroxide values obtained from both tiger nut at moisture content of 13%
toasted for 30 min at 90°C toasting temperature. The peroxide value was observed
to increase as toasting time and toasting temperature increased but decreased as
the moisture level increased at 50°C and 70°C toasting temperatures, although
an increase was observed as the moisture level increases at 90°C toasting
temperature. Analysis of variance indicated a significant effect of the
moisture level, toasting time, toasting temperature, and variety on the
peroxide value at 95% confidence level. The peroxide value range obtained in
this research is within the acceptable limit of <10 meqO2/Kg
peroxide value for edible oil as reported for olive oil, cotton seed oil, soya
beans oil, maize oil, palm oil among several others published by [27] fats and oil regulations and the [28]. [26] reported the same trend in their study
of roasted soybean. The result obtained in this study is different from the
range of 0.71-1.36 meqO2/Kg reported by [10]
and 15.76 meqO2/Kg reported by [29]
for tiger nut oil peroxide value and this difference could be because of
the different oil extraction, drying methods applied and the differences in
moisture content applied in this study. Because of the high toasting
temperatures used, the thermal oxidation of the oils is triggered, leading to a
higher range of peroxide value than the one reported by [10]. The results corresponded with the peroxide value reported
by [24] for Almond seed oil and Soybean
seed oil reported by [33].
Table 3. Average results of the tiger nut oil Peroxide value (PV) investigated as influenced by variety, moisture level,
toasting temperature, and toasting time.
|
Moisture Level (%) |
Toasting
Temperature (°C) |
Toasting Time (min) |
Brown Tiger nut PV (meq O2/Kg) |
Standard Deviation |
Yellow Tiger nut PV (meq O2/Kg) |
Standard Deviation |
|
7 |
50 |
10 |
2.33aaa |
0.0321 |
2.61aaa |
0.0321 |
|
20 |
2.74aab |
0.0100 |
2.79aab |
0.0115 |
||
|
30 |
2.92aac |
0.0058 |
3.32aac |
0.0586 |
||
|
70 |
10 |
2.38aba |
0.0153 |
3.35aba |
0.0954 |
|
|
20 |
2.54abb |
0.0208 |
3.78abb |
0.0600 |
||
|
30 |
2.90abc |
0.0208 |
3.96abc |
0.0153 |
||
|
90 |
10 |
3.35aca |
0.0100 |
3.18aca |
0.1400 |
|
|
20 |
3.77acb |
0.0265 |
3.81acb |
0.0231 |
||
|
30 |
3.89acc |
0.0400 |
3.75acc |
0.0100 |
||
|
10 |
50 |
10 |
2.23baa |
0.0208 |
2.42baa |
0.0000 |
|
20 |
2.66bab |
0.0115 |
2.82bab |
0.0700 |
||
|
30 |
2.93bac |
0.0643 |
3.06bac |
0.0153 |
||
|
70 |
10 |
2.33bba |
0.0100 |
3.95bba |
0.0100 |
|
|
20 |
2.35bbb |
0.0100 |
3.97bbb |
0.0200 |
||
|
30 |
2.66bbc |
0.0173 |
4.27bbc |
0.0737 |
||
|
90 |
10 |
3.85bca |
0.0058 |
3.56bca |
0.0305 |
|
|
20 |
3.95bcb |
0.0153 |
4.07bcb |
0.0473 |
||
|
30 |
4.35bcc |
0.0100 |
3.95bcc |
0.0058 |
||
|
13 |
50 |
10 |
2.18caa |
0.0252 |
2.25caa |
0.0416 |
|
20 |
2.50cab |
0.0115 |
2.62cab |
0.0252 |
||
|
30 |
2.71cac |
0.0200 |
2.95cac |
0.0058 |
||
|
70 |
10 |
2.26cba |
0.0115 |
4.01cba |
0.0436 |
|
|
20 |
2.26cbb |
0.0608 |
4.31cbb |
0.0458 |
||
|
30 |
2.62cbc |
0.0100 |
4.48cbc |
0.0404 |
||
|
90 |
10 |
3.98cca |
0.0305 |
3.74cca |
0.0058 |
|
|
20 |
4.35ccb |
0.0173 |
4.29ccb |
0.0436 |
||
|
30 |
4.52ccc |
0.0300 |
4.45ccc |
0.0058 |
||
|
Note: The first, second and third letters
in the superscript represents the effects of moisture content, toasting
temperature and toasting time on oil quality respectively. Means with the same superscript are not
significantly different along the same column
|
||||||
3.4 Effects of
variety, moisture level and toasting time on Refractive Index
The experimental samples
displayed a refractive index range of 1.45-1.47 as reported in Table 4, and the
brown tiger nut sample at moisture level 7% toasted for 20 min at 50°C toasting
temperature is observed to have the least value while the yellow tiger nut
sample at moisture level 13% toasted for 10 min at 50°C toasting temperature
alongside brown tiger nut sample at moisture level 13% toasted for 30 min at
70°C toasting temperature all have the highest refractive index values. The
refractive index value was observed to decrease as the toasting temperature and
toasting time increased but increases
as the moisture level increased from 7% to 13% respectively. Analysis of
variance reported a significant effect of moisture level, variety, toasting
time and toasting temperature on the refractive index value at p<0.05.
Most of the range of refractive index values obtained in this research is
within the acceptable range of refractive index 1.4677-1.4707 as published by [27, 28] fats and oil regulations while some are
little below the acceptable value which could be because of the influence of
the pre-treatment given to the tiger nut before oil extraction or because of
its unrefined characteristics. [29] reported 1.48 refractive index for
tiger nut oil extracted mechanically but without any treatment. The
refractive index value of the tiger nut oil is within the report on Refractive
index of Soybean seed oil, castor seed oil, Pumpkin seed oil, and almond seed
oil by [24, 33-37] reported a trend similar to the
observation in this study.
Table 4. Average results of the tiger nut oil Refractive index (RI) investigated as influenced by variety, moisture level,
toasting temperature and toasting time.
|
Moisture Level (%) |
Toasting Temperature
(°C) |
Toasting Time (min) |
Brown Tiger nut RI |
Standard Deviation |
Yellow Tiger nut RI |
Standard Deviation |
|
7a |
50c |
10b |
1.47acb |
0.001 |
1.46acb |
0.001 |
|
20a |
1.45aca |
0.024 |
1.46aca |
0.001 |
||
|
30a |
1.46aca |
0.000 |
1.46aca |
0.001 |
||
|
70b |
10 |
1.47abb |
0.001 |
1.45abb |
0.001 |
|
|
20 |
1.46aba |
0.000 |
1.45aba |
0.001 |
||
|
30 |
1.47aba |
0.001 |
1.45aba |
0.001 |
||
|
90a |
10 |
1.45aab |
0.001 |
1.45aab |
0.001 |
|
|
20 |
1.45aaa |
0.001 |
1.45aaa |
0.001 |
||
|
30 |
1.45aaa |
0.001 |
1.45aaa |
0.012 |
||
|
10b |
50 |
10 |
1.47bcb |
0.001 |
1.47bcb |
0.001 |
|
20 |
1.47bca |
0.000 |
1.47bca |
0.001 |
||
|
30 |
1.47bca |
0.001 |
1.47bca |
0.001 |
||
|
70 |
10 |
1.47bbb |
0.000 |
1.46bbb |
0.001 |
|
|
20 |
1.47bba |
0.000 |
1.46bba |
0.001 |
||
|
30 |
1.47bba |
0.001 |
1.46bba |
0.003 |
||
|
90 |
10 |
1.46bab |
0.000 |
1.46bab |
0.001 |
|
|
20 |
1.46baa |
0.001 |
1.45baa |
0.001 |
||
|
30 |
1.46baa |
0.001 |
1.45baa |
0.002 |
||
|
13c |
50 |
10 |
1.47ccb |
0.001 |
1.47ccb |
0.001 |
|
20 |
1.47cca |
0.000 |
1.47cca |
0.001 |
||
|
30 |
1.47cca |
0.001 |
1.47cca |
0.000 |
||
|
70 |
10 |
1.47cbb |
0.001 |
1.46cbb |
0.003 |
|
|
20 |
1.47cba |
0.000 |
1.46cba |
0.001 |
||
|
30 |
1.47cba |
0.001 |
1.46cba |
0.002 |
||
|
90 |
10 |
1.46cab |
0.003 |
1.46cab |
0.002 |
|
|
20 |
1.46caa |
0.004 |
1.46caa |
0.000 |
||
|
30 |
1.46caa |
0.002 |
1.46caa |
0.002 |
||
|
Note: The first, second and third letters
in the superscript represents the effects of moisture content, toasting
temperature and toasting time on oil quality, respectively. Means with the same superscript in the same
column are not significantly different
|
||||||
3.5 Effects
of variety, moisture level and toasting time on Relative Density
The relative density of the various samples experimented is
represented in Table 5 and it ranged from 0.91-0.98. The least value of the
relative density was obtained from the yellow tiger nut sample at 7% moisture
level toasted at 20 min while the yellow tiger nut at 13% moisture level
toasted at 10 min had the highest value. The relative density of the samples
showed an increase simultaneously as the moisture level increased but decreased
as the toasting time and temperature increased. Analysis of variance revealed a
significant effect of the moisture level, toasting time and variety on the
specific gravity or relative density value at 5% probability error. Majority of
the relative density of the experimented samples falls within the acceptable
value of specific gravity value for olive oil, soya beans oil and cotton seed
oil as published by [27, 28] fats and oil
regulations, while little is above the maximum acceptable value which was
because of the influence of the pre-treatment given to the tiger nut before oil
extraction. The result reported is in line with the relative density reported
for Castor seed oil, Pumpkin seed oil, Almond seed oil by [35], and [24] respectively.
In the reported studies [38-40] on Roasted
coconut oil, roasted peanut oil and sesame oil all reported a similar trend of
observation. The relative density of the tiger nut oil was less dense than that
of water which means, the oil is light and unsaturated which does not have an
extreme lubricating potential. This is an indication tiger nut oil will be a
good ingredient in the production of creams since the oil can easily be
distributed or applied on human skin.
Table 5. Average results of the tiger nut oil Relative Density (RD) investigated as influenced
by variety, moisture level, toasting temperature, and toasting time.
|
Moisture Level (%) |
Toasting
Temperature (°C) |
Toasting Time (min) |
Brown Tiger nut RD |
Standard Deviation |
Yellow Tiger nut RD |
Standard Deviation |
|
7 |
50 |
10 |
0.94ccb |
0.001 |
0.93ccb |
0.001 |
|
20 |
0.96cca |
0.002 |
0.98cca |
0.002 |
||
|
30 |
0.94cca |
0.002 |
0.97cca |
0.002 |
||
|
70 |
10 |
0.94cbb |
0.000 |
0.93cbb |
0.001 |
|
|
20 |
0.95cba |
0.001 |
0.95cba |
0.002 |
||
|
30 |
0.96cba |
0.002 |
0.95cba |
0.001 |
||
|
90 |
10 |
0.93cab |
0.001 |
0.93cab |
0.001 |
|
|
20 |
0.94caa |
0.002 |
0.93caa |
0.002 |
||
|
30 |
0.94caa |
0.001 |
0.95caa |
0.001 |
||
|
10 |
50 |
10 |
0.93bcb |
0.000 |
0.92bcb |
0.001 |
|
20 |
0.95bca |
0.001 |
0.97bca |
0.001 |
||
|
30 |
0.92bca |
0.000 |
0.96bca |
0.002 |
||
|
70 |
10 |
0.94bbb |
0.001 |
0.92bbb |
0.001 |
|
|
20 |
0.95bba |
0.001 |
0.94bba |
0.001 |
||
|
30 |
0.94bba |
0.002 |
0.94bba |
0.001 |
||
|
90 |
10 |
0.92bab |
0.001 |
0.92bab |
0.001 |
|
|
20 |
0.93baa |
0.001 |
0.93baa |
0.002 |
||
|
30 |
0.93baa |
0.001 |
0.95baa |
0.001 |
||
|
13 |
50 |
10 |
0.92acb |
0.001 |
0.91acb |
0.000 |
|
20 |
0.94aca |
0.001 |
0.94aca |
0.004 |
||
|
30 |
0.91aca |
0.001 |
0.93aca |
0.001 |
||
|
70 |
10 |
0.93abb |
0.001 |
0.92abb |
0.001 |
|
|
20 |
0.94aba |
0.002 |
0.94aba |
0.002 |
||
|
30 |
0.92aba |
0.002 |
0.93aba |
0.001 |
||
|
90 |
10 |
0.92aab |
0.000 |
0.92aab |
0.002 |
|
|
20 |
0.93aaa |
0.002 |
0.92aaa |
0.001 |
||
|
30 |
0.92aaa |
0.001 |
0.92aaa |
0.001 |
||
|
Note: The first, second and third letters in the superscript represents the effects of
moisture content, toasting temperature and toasting time on oil quality,
respectively. Means with the same
superscript in the same column are not significantly different. |
||||||
4. Conclusions
This study evaluated
the quality of tiger nut oil as affected by the processing conditions and
variety. Free fatty acid (FFA), iodine value (IV), peroxide value (PV),
refractive index (RI) and relative density (RD) of two varieties of tiger nut
oil extracted mechanically were evaluated to ascertain the effects of variety
(brown and yellow) and processing conditions namely; moisture content (7 %, 10
% and 13 %), toasting temperature (50 ˚C, 70 ˚C and 90 ˚C) and toasting time
(10, 20 and 30 min) on the extracted oil. The brown tiger nut was observed to
have a better quality in terms of the indices measured than the yellow tiger
nut oil. And the treatment with the best oil quality was the brown tiger nut at
7 % moisture content, 50 ˚C toasting temperature and 10 min toasting time. The
FFA, IV, PV, RI and RD for both varieties of tiger nut oil under the ultilised
processing conditions were recorded to be in the range of 0.22 - 0.49 (%),
52.62 - 80.20 (mg/100g), 2.18 - 4.52 (meqO2/Kg), 1.45 - 1.47 and
0.91 - 0.98 respectively. It was observed that tiger nut oil quality for the
two varieties reduced as the moisture level, toasting temperature and time
increased. The results data shows that both variety and processing conditions
have significant impact on the evaluated quality of tiger nut oil at P≤0.05.
The tiger nut oil quality from both varieties investigated are within the range
acceptable for vegetable oils and can be considered a viable
alternative to common vegetable oil in food related applications.
Authors’ contributions
Idea conception, experimental design, experimentation,
manuscript, writing of the original draft, P.A.O.; Idea conception, project
supervision, review and editing, A.K.A.; Idea conception, writing-review and
editing, M.O.O.; Experimentation, review and statistical analysis, T.O.O.;
Project supervision, experimentation, writing–review and editing, A.A.A.
Acknowledgements
We acknowledge the Tertiary Education Trust Fund (TETFund)
of Nigeria for sponsoring the bench work at the University of Kentucky to complete
some aspects of this study. We also acknowledge the University of Kentucky and
the Department of Biosystems and Agricultural Engineering for giving access to
the Food Engineering Laboratory where the oil quality properties analysis was
carried out. Finally, we appreciate the Department of Agricultural and
Environmental Engineering, University of Ibadan, and the University of
Agriculture Makurdi, Nigeria for their technical support in completing this
study on Tiger nut and its oil.
Funding
This
study was partly funded by Tertiary Education Trust Fund of Nigeria
Conflicts of interest
The authors declare there are no known competing financial
interests or personal relationships that could have appeared to influence the
work reported in this paper.
References
1.
El-Din, D.M.E. Tiger Nuts: A revival of an ancient Egyptian
plant, J. Res. Centr. Egypt. 2021,
57(1), 57-74.
2.
Awulu, J.O.; Omale, PA.; Omadachi, J.O. Characterization of tiger nut oil extracted
using mechanical and chemical methods. J. Sci. Multidisci. Res. 2018, 10(2),
13-25.
3.
Bazine,
T.; Arslanoglu, F. Tiger nut (Cyperus esculentus): morphology, product,
uses and health benefits. Black Sea J. Agric. 2020, 3, 324-328.
4.
Balami,
A.A.; Birma, M.; Dauda, S. M.; Adeboye, S.E. Engineering properties of tiger
nut relative to the design of a cleaning and sorting machine. In Proceedings of
the 2nd International Conference on Advances in Applied Science and
Environmental Engineering. Kuala Lumpur, Malaysia, 20-21 December, 2014.
5.
Omale,
P.A.; Iyidiobu, B.N.; Ibu, E.J. Effect of drying temperature on the nutritional
quality of tiger nut (Cyperus Esculentus L). Int. J. Engr. Appl. Sci. Tech. 2020, 4(9),
399-403.
6.
Aremu,
M.O.; Ibrahim, H.; Bamidele, T.O. Physicochemical characteristics of the oils
extracted from some Nigerian plant foods-A review. Chem. Engr. Res. 2015, 32,
36-52.
7.
Chu,
C.C.; Nyam, K.L. Seed Oil: Sources, Properties and Recovery, In Recent Advances
in Edible Fats and Oils Technology.
Springer, Singapore. 77-100, 2022.
8.
Makareviciene, V.; Gumbyte, M.; Yunik, A.;
Kalenska, S.; Kalenskii, V.; Rachmetov, D.; Sendzikiene, E. Opportunities for
the use of chufa sedge in biodiesel production. Ind. Crops Prod. 2013, 50,
633-637.
9.
Kizzie-Hayford,
N.; Dabie, K.; Kyei-Asante, B.; Ampofo-Asiama, J.; Zahn, S.; Jaros, D.; Rohm,
H. Storage temperature of tiger nuts (Cyperus esculentus L) affects
enzyme activity, proximate composition and properties of lactic acid fermented
tiger nut milk derived thereof. LWT. 2021, 137, 110417.
10.
Zhang,
Z.S.; Jia, H.J.; Li, X.D.; Liu, Y.L.; Wei, A.C.; Zhu, W.X. Effect of drying
methods on the quality of tiger nuts (Cyperus esculentus L.) and its
oil. LWT. 2022, 167, 113827.
11.
Ezebor, F.; Igwe, C.C.; Owolabi,
F.A.T.; Okoh, S.O.; Comparison of the physico-chemical characteristics,
oxidative and hydrolytic stabilities of oil and fat of Cyperus esculentus L.
and Butyrospermum parkii (Shea nut) from Middle-Belt States of Nigeria. Int. J.
Fd. Sci. Tech. 2013, 48(12), 2588-2595.
12.
Ezeh,
O.; Michael, H.G.; Niranjan, k. Tiger nut oil (Cyperus esculentus L.): A
review of its composition and physio-chemical properties. Eur. J. Lip. Sci.
Tech. 2014, 116(6), 783-794.
13.
Yali,
Y.U.; Xiaoyu, L.U.; Zhang, T.; Zhao, C.; Guan, S.; Yiling, P.U.; Gao, F. Tiger
Nut (Cyperus esculentus L.): nutrition, processing, function and
applications. Foods. 2022, 11(4), 601.
14.
ASABE S352. Moisture Measurement-Unground Grain
and Seeds. Published by ASABE, United State, April 1988.
15.
Akinoso,
R. Effects of moisture content, roasting duration and temperature on yield and
quality of Palm kernel (Elaeis guineensis) and Sesame (Sesamum
indicum) oils. Ph.D. Thesis, University of Ibadan, Ibadan, Nigeria. 2006.
16.
Ogunlade,
C.A. Optimization and characterization of mechanically expressed oil from African
oil bean (Pentaclethra Macrophylla Benth) kernels. Ph.D. Thesis,
University of Ibadan, Ibadan, Nigeria 2018.
17.
AOAC.
Official Method 940.28. Fatty
Acids (Free) in Crude and Refined Oils. Association of Official Analytical
Chemists, Washington D.C. Titration Method. 22nd Edn., 2023.
18.
Ogori,
A.F.; Nina, G.C.; Ukeyima, M.T. Quality characteristics of stored varieties of
tiger nut oil. Food. Life. 2021, 1, 29-34.
19.
AOAC. Official Method 993.20. Iodine value of Fats and Oils. Wijs
(cyclohexane-Acetic Acid Solvent) Method. Association of Official Analytical
Chemists, Washington D.C. Final Action. 22nd Edition, 2023.
20.
ISO
3961. Animal and vegetable fats and oils - determination of iodine value. 8th
Edn., International Standards Organization, Geneva, Switzerland, 2018
21.
ISO
3960. Animal and vegetable fats and oils-determination of peroxide value –iodometric
(visual) endpoint determination. International Standards Organization, Geneva,
Switzerland, 2017.
22.
ISO
6320:2017 Animal and vegetable fats and oils - determination of refractive
index. International Standards Organization, Geneva, Switzerland, 2017.
23.
Ogunsuyi, H.O.; Daramola, B.M. Evaluation of almond (Prunus amygdalus) seed
oil as a viable feedstock for biodiesel fuel. Int. J.
Biotech. Res. 2013, 1(8), 120-127.
24.
Afuape, Z.O.; Oke, E.
K.; Idowu, M.A.; Olorode, O.O.; Omoniyi, S.A.
Physical and chemical properties of tigernut oil as influenced by variety and
method of extraction. The Annals of the University Dunarea de Jos of Galati
Fascicle VI – Fd. Tech. 2021, 45(1), 129-140.
25.
Mohammed,
M.I.; Hamza, Z.U. Physicochemical properties of oil
extracts from Sesamum Indicum L. seeds grown in Jigawa State–Nigeria. J.
Appl. Sci. Environ. Managt. 2008, 12(2), 99–101.
26.
Chen, H.; Zhou, Y.; Liu, J. Effects of
roasting time on the quality of soybean oil extracted from roasted soybeans. Food.
Sci. Nutr. 2021, 9(5), 2665-2673.
27.
National
agency for food and drug administration and control (NAFDAC), Fat and oil
regulations in Nigeria, 2019.
28.
Codex standard for named vegetable oils
(CODEX-STAN 210-1999). International food standard, guidelines, and code of
practice, 1999.
29.
Guo,
T.; Wan, C.; Huang, F.; Wei, C. Evaluation of quality properties and
antioxidant activities of tiger nut (Cyperus esculentus L.) oil produced
by mechanical expression or/with critical fluid extraction. LWT. 2021, 141,
110915.
30.
Silvia, M.; Federica, P.; Vito, V.;
Hanna, C.; Maria, F.C.; Santina, R. Effects of different roasting conditions on
physical-chemical properties of Polish hazelnuts (Corylus avellana L.
var. Katalonski). LWT. 2017, 77, 440-448.
31.
Oomah, B.D.; Kenaschuk, E.O.; Mazza,
G.; Liang, J. Roasting effects on the quality and stability of canola oil. J.
Am. Oil Chem. Soci. 2010, 87(4), 389-397.
32.
Nangbes,
J.G.; Nvau, J.B.; Buba, W.M.; Zukdimma, A.N. Extraction and characterization of
castor (Ricinus Communis) seed oil. The Int. J. Engr. Sci. 2013, 2(9),
105–109.
33.
Javidipour, H.; Erinç,
A.; Baştürk, A.; Tekin, A. Oxidative changes in hazelnut,
olive, soybean, and sunflower oils during microwave heating. Int. J. Food. Prop.
2016, 20(7), 1582-1592.
34.
Akpan,
U.G.; Jimoh, A.; Mohammed, A.D. Extraction, characterization and modification
of castor seed oil. Leonardo J. Sci. 2010, 8, 43-52.
35.
Bwade,
K.E.; Aliyu, B.; Kwaji, A.M. Physicochemical properties of pumpkin seed oil
relevant to bio-diesel production and other industrial applications. Int. J.
Engr., Bus. Enterp. Applic. 2017, 4(1), 72–78.
36.
Srinivas, K.; Rao, B.D.; Raghavarao, K.S.
Influence of roasting time on the physico-chemical properties of sesame oil
extracted from roasted seeds. J. Food Meas. Charact. 2021, 15(1), 293-300.
37.
Willard, B.N.; Yufei, H.; Kingsley, M.;
Caimeng, Z. Effect of roasting temperatures and times on test parameters used
in determination of adequacy of soybean processing. Adv. J. Food. Sci. Tech.
2017, 13(1), 22-28.
38.
Bolarinwa, I.F.; Alamu, O.T.; Olawale,
A.K. Effects of moisture content on the physical properties of coconut oil
extracted from roasted kernels. J. Food. Meas. Charact. 2019, 13(4), 2554-2562.
39.
Raju, P.S.; Annapurna, Y.; Sharma, R.
Effect of roasting temperature on the quality characteristics of groundnut oil
extracted from roasted peanuts. J. Food. Meas. Charact. 2019, 13(2), 1060-1070.
40.
Singh, A.K.; Singh, R.; Singh, N.;
Saxena, D.C. Effects of roasting temperature on the quality of sesame oil
extracted from roasted sesame seeds. J. Food. Meas. Charact. 2021, 15(1),
415-423.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Tiger nuts (Cyperus esculentus) are underutilized edible oil seeds
cultivated across Africa with potential as a source of healthy vegetable oil.
It is rich in oil with about 22.14 – 44.92% of the dry weight. This study’s main
aim was to evaluate the quality of tiger nut oil and determine the best variety
and processing conditions for producing high-quality from it to meet the
increasing demand for healthier and more sustainable vegetable oils. Applying a completely randomized
design, oil was extracted
mechanically using an oil expeller and the impact of variety (yellow and brown), moisture level (7%, 10% and 13%), toasting temperature (50 °C, 70 °C and 90 °C) and toasting time (10, 20 and 30
min) were investigated. Using standard procedures, five oil quality parameters
were determined, namely free fatty acid (FFA),
iodine value (IV), peroxide value (PV), refractive index (RI) and
relative density (RD). The result shows a range of 0.22 - 0.49% for FFA, 52.62 – 80.20 (mg/100g) for IV, 2.18-
4.52 (meqO2/Kg) for PV, 1.45 - 1.47 for RI,
and 0.91 - 0.98 for RD. FFA and PV
all increased as the moisture level, toasting time and toasting temperature increased,
whereas the IV decreases. The RI and RD values
decreased as the toasting temperature and toasting time increased but
increased as the moisture level increased. ANOVA
indicates that variety, moisture
level, toasting temperature and toasting time have
significant effects on the FFA, IV,
PV, RI and RD at P ≤0.05. The results show that the best oil quality was achieved at 7 % moisture content, 50 °C
toasting temperature and 10 min toasting time for both varieties. The
brown tiger nut has more quality oil than the yellow Tiger nut. The tiger nut oil has comparable characteristics
common to vegetable oils used in food related
applications and can be considered a viable
alternative to common vegetable oils for food preparation. Findings from this study will help tiger nut
oil processors to make the right processing pretreatment before oil extraction.
Abstract Keywords
Free fatty acid, iodine value, peroxide value,
tiger nut, tiger nut oil
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


