Research Article
Dur-e-shahwar Sattar*
Dur-e-shahwar Sattar*
Corresponding
Author
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: dsattar@bzu.edu.pk, Tel: +923337754842
Muhammad Faisal Jehanzeb Khan
Muhammad Faisal Jehanzeb Khan
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: faisalmniazi@gmail.com
Rani Aliha Mushtaq
Rani Aliha Mushtaq
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: alihamushtaq786@gmail.com
Muhammad Ahsan Javed
Muhammad Ahsan Javed
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: mahsanjaved356@gmail.com
Maryam Jalal Ud Din
Maryam Jalal Ud Din
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: bukharimaryam007@gmail.com
Hira Chaudhary
Hira Chaudhary
Department of Food Science and Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, 60700, Multan, Pakistan.
E-mail: hirach494@gmail.com
Abstract
The
research investigated the restoration of spoil sorghum by utilizing neem
extract to create biodegradable films. Sorghum is one of the most important cereal
crops that frequently spoils and causes a lot of waste. In an effort to lessen
the waste, the research was to make biodegradable, eco-friendly films that
would increase the worth and cut down on waste. The antibacterial and
antioxidant qualities of neem extract were added to the sorghum-based film
matrix. The resulting films were assessed for biodegradability, antibacterial
effectiveness, and mechanical, thermal, and barrier qualities. The results suggested
that the incorporation of 7.5% neem extract into sorghum starch films improved
their biodegradability and also color properties. F7.5% shows more
stable result of moisture content while F15% causes drying effect
which results in reduction of film moisture content. The pH values in F7.5%
and F15% remain stable indicated that neem extract effectively
inhibits microbial activity and prevents acidification.
Abstract Keywords
Spoiled
sorghum starch, neem extract, biodegradable films, film solubility, water
vapour permeability, mechanical test, shelf life of coriander leaves.
1. Introduction
Sorghum (Sorghum bicolor L.) belongs to the family Gramineae which is the fifth-most significant cereal crop after rice, wheat, corn, and barley. Sorghum is the primary food grain consumed by about 750 million people in the semi-arid tropical regions of Africa, Asia, and Latin America. Small-scale subsistence farmers make up the majority of producers [1]. They have limited access to production inputs such as fertilizer, herbicides, improved seeds (hybrids or variations), healthy soil, water, and better financing options for buying them. Globally, it is a significant source of food, feed, and forage [2]. The primary factor driving the increase in demand for sorghum as feed is worldwide production and international commerce. Additionally, it has been utilized in the brewing of alcohol. The entire plant is utilized for fodder and hay or silage. However, sorghum grains could be spoiled easily due to extreme temperature, relative humidity, grain moisture contents, and association of microflora with sorghum grain stored in underground pits under different agro-ecological conditions [3]. It can also be contaminated by mold growth. Grain infection starts at the base, close to the pedicel, and interferes with grain filling, causing the black coating to appear too soon. Smaller seeds are produced as a result, which lowers yields and causes seed dormancy [4]. Spoilage fungus develops at a moisture level of 15–19%, which causes a notable increase in respiratory activity. As a result of the successive colonization of thermophilous fungi, there is a rise in temperature and occasionally spontaneous heating [5].
Neem leaves are one of the best ways to
prevent the production of aflatoxin (Azadirachta indica A. Juss). They
are considered as biopesticides and natural anti-fungi agents because natural
biopesticides generated from plants may biodegrade and have few adverse
effects, they are a highly safe product to use [6].
The active components in neem leaf extract, azadirachtin, solanine, melantriol,
and nimbin, are said to have pesticidal properties [7].
Compounds found in neem leaves, including azadirachtin, solanine, nimbin, and
nimbidin act as invaders of cell development and have the ability to cause
fungal cell death [8].
Thus, the incorporation of neem extract in the spoiled sorghum starch to
produce biodegradable film would be an innovative idea for the food waste
management. Biodegradable films are made with the goal of displacing polyethylene
film, which is utilized for a variety of applications such as industrial films,
product packaging, and organic waste collecting bags [9].
Comparing these materials to conventional non-biodegradable plastics revealed
superior qualities. They can withstand wet and heated organic materials for
several weeks or even months without experiencing any physical changes. This
makes the composting program more flexible and also works well as a substitute
for the present films used for product packaging, transportation, and storage
and are fully biodegradable [10].
In addition, they are composed of
sustainable biomaterials (polyester generated from corn dextrose), do not
include polyethylene, and do not leave residues after composting [11].
In light of these circumstances, the
aim of the present study was to utilize spoiled sorghum starch to form
biodegradable film with the incorporation of various concentrations of neem
extract. Various physical parameters such as film thickness, film moisture
analysis, film solubility and water permeability were determined. Moreover,
physicochemical quality parameters such as color profile, mechanical testing
(tensile strength and percent elongation), and biodegradable tests were performed.
Shelf-life assessment of composite coriander leaves on the basis of chlorophyll
content, microbial load, pH, titratable acidity and moisture content was examined
to understand the extension of shelf life in coriander leaves.
2. Materials and methods
2.1.
Procurement of raw material
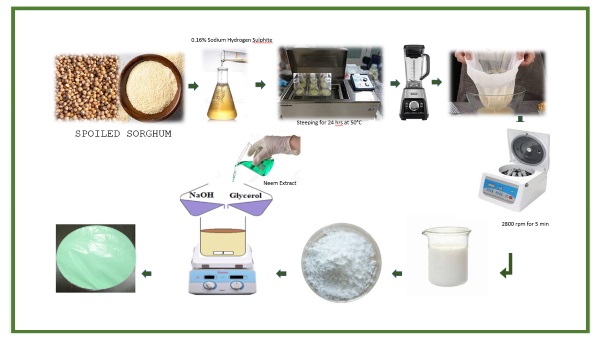
This research was carried out in food analysis laboratory of Faculty of Food Science and Nutrition, BZU, Multan. Spoiled Sorghum were collected from the dump site of Multan. They were cleaned and used for further analysis.
2.2. Sorghum cleaning
Spoiled sorghum was cleaned and foreign materials were removed manually followed by washing sorghum with distilled water at least five times. For a better result, 10 mL of SiO2 solution were added to a 2kg sample for 24 hours at 50 °C. Again, the samples were washed, dried and kept at 0 °C in the freezer to safe from further deterioration.
2.3.
Isolation of starch from spoiled
sorghum grains
The
procedure was used to isolate sorghum starch. Grains were washed and steeped
for 24 hours at 50 °C in a solution of 0.2% SO2 and 0.5% lactic
acid. The steeping solution was completely removed from the grains by giving
them two thorough washes. After that, the sorghum kernels were roughly
pulverized and put through a 5-minute, 8000 rpm blender cycle. However, blending
was stopped for two minutes after every minute of blending to avoid continuous
mixing for five minutes, which could cause the starch to gelatinize. After applying a 0.5 M NaOH solution to get the resulting slurry's pH to
8.5, it was let to stand for 30 minutes. Next, it passed through a stack of
wire mesh sieves with sizes of 80, 170, and 270, arranged from top to bottom.
After two minutes of blending, the leftovers from the sieves were sieved. This
procedure was carried out twice. Following this, the starch slurry was
homogenized for 60 seconds at 4 ºC using a homogenizer (Polytron PT 2100,
Kinematica Inc., Switzerland). A spatula was used to scrape off the protein
layer. After washing with water, the sediment starch was homogenized once more
in order to eliminate any remaining protein. The starch was then rinsed and
dried for 48 hours at 45 ºC in a forced air oven. Before usage, it was
subsequently kept in a sealed plastic bag [12].
2.4. Neem extract preparation
Neem leaves
were detached from the stems and were washed and dried for five days in an oven
at 45 °C. Dried leaves were pulverized with a grinder. An amber-colored bottle
held the sample of leaf powder.
Extraction of neem extract was done by adding 100 mL of ethanol to 20 grams of
neem powder (5:1) for 24 hours at 37 ℃ followed by sieving through filter
paper and then stored at 4 ℃ [13].
2.5.
Starch film preparation
Starch films were prepared by adding
three grams of starch in 96 mL of warm distilled water with continuous
stirring. After the starch was gelatinized
completely at 90 ℃ cool to room temperature, followed by adding 30 percent of
glycerol and active agent neem extract (7.5 and 15% respectively), the solution
was stirred continuously for 15 minutes on a magnetic stirrer. The solution was
poured on a petri plate and kept at 45 in the oven for 24 hours. After cooling,
films were peeled off and stored for further analysis [14].
2.6.
Determination of physical parameters of films
2.6.1. Film thickness measurement
Using a
digital micrometer screw gauge (Insize, Loganville, USA), the thickness of film
samples was measured with a precision of 0.01 mm. Each film sample had
measurements made at ten different random places, and the average value was
given in millimeters.
2.6.2. Film moisture analysis
A (2 x 2) cm square piece of film was dried to 0%
moisture content in a moisture analyzer (Radwag MAC 50/1, Poland) to determine
the moisture content of sorghum starch films. The formula was utilized to
ascertain the moisture content of the films based on three separate readings [15].
2.6.3. Film solubility
With a few minor modifications, methodology was applied to ascertain the solubility of films. After drying, film samples were submerged in 50 milliliters of distilled water and left in a shaking water bath at 25 degrees Celsius for a whole day. The resulting moist film samples were dried in a moisture analyzer to 0% moisture content after being filtered through Whatman No. 1 filter paper [15]. The formula for film solubility was:
Where
m1 = weight of initially dried film
m2 = weight of soaked film after drying
2.6.4.
Determination of Water Vapor Permeability
ASTM
(ASTM) Method E96 with some modifications was employed to measure the water
vapour permeability (WVP) of films. A 0 % RH permeation cell was maintained
using medium granular silica gel blue and the film to be tested was sealed to
the mouth of it. This cell was then placed inside the desiccator with a saturated
solution of sodium chloride to maintain the RH to 75%. The RH of the desiccator
was always greater than that of the permeation cell. After about 2 hours or
more when the steady state was attained, the water vapor transport rate (WVTR)
was calculated through the weight gain of the film. The measurements were taken
at 1 hour intervals and the graph was plotted against weight gain and time. The
linear regression method was employed to find out slope. WVTR was calculated by
dividing slope and transfer area (m²). Film thickness was measured after
completion of the permeation testing. The equation reported by Mali et al.
(2004) was used to calculate the WVP (gm-1s-1Pa-1):
WVP
= WVTRS (𝑅1−𝑅2) × D
Where,
S is the saturated vapor pressure of water (Pa),
R1 and R2 are the percent RH
of the permeation cell and desiccator at 25˚C, respectively, and D is the
thickness of the film (m) [15].
2.7. Determination of color profile
of films
The
color profile was determined through the utilization of a colorimeter, namely
the Hunter Lab Color Quest colorimeter. The variables a* (indicating
greenness/redness), L* (representing darkness/lightness), and b* (denoting
blueness/yellowness) were employed to define the color profile [15].
2.8. Determination of mechanical
properties of film
The Universal Testing Machine (Zwick/
Roell, GmbH & Co., D-89079 Ulm, Germany) UTM was used to assess the
mechanical properties of film. The mechanical properties of the films were
evaluated by determining the tensile strength and % elongation of the films.
Prior to testing, films were conditioned for 7 days at 55% RH and at 25˚C. Films
were cut into 3 cm wide and 10 cm length wise strips. Film strips were then
held between two clamps of UTM in such a manner that one end of the strip was
attached to the moving arm and the other was fixed on the platform. The
operation conditions were as follows: a load cell, 1 kilo Newton; grip to grip
separation, 10 cm; and crosshead speed, 50 mm/min; force shutdown threshold,
30%. The upper clamp stretched the film strip till it ruptured from the centre.
The mechanical properties of each film were measured in ten replicates. The
maximum 95 force required to rupture Fmax was measured in MPa and
elongation in % was directly calculated by means of test Xpert software.
2.9. Determination
of biodegradability of film
Biodegradability
measures the film’s resistance to degrading microorganisms, soil, moisture,
temperature, and other physicochemical factors. Biodegradability tests were
conducted using the weight-loss method. Each film sample (4 cm × 4 cm size) was
buried in a plastic jar at around 12 cm depth in the soil and maintained at
room temperature for 30 days. The
recovered test samples were rinsed under running water to remove the soil
residue from the surface and dried at 80 °C in an oven. The initial weight (W5)
and final weight (W6) before and after testing were recorded. The weight loss
obtained after the testing relates to each film sample’s biodegradability level
[16].
2.10.
Shelf life assessment of coriander leaves
2.10.1.
Total chlorophyll content
To measure the amount of chlorophyll,
0.5 g of the sample was pulverized in a pestle and mortar with 3 mL of pure
acetone and then 5 mL of 80% acetone [17].
After centrifuging the solution at 4000 rpm for 10 min, the supernatant’s
volume was reduced to 10 mL by adding 80% acetone. Using a LAMBDA 365 UV/vis
Spectrophotometer (PerkinElmer, Waltham, MA), the absorbance was measured at
663 and 645 nm. The following equation was used to determine the total
chlorophyll content:
Total chlorophyll content (mg/g) = (20.29𝐴663+8.05𝐴645) ×𝑉/𝑊×1000
Where, V stands for solution volume and
W stands for sample weight.
2.10.2.
Total microbiological load
Using the total plate count approach,
the total number of microbes was calculated. For the 10–1 dilution, 1 g of
coriander leaves and 9 mL of a sterile physiological solution (0.85% NaCl) were
used to homogenize the mixture and prepare the sample solution. Subsequently, a
serial dilution was performed to reach a concentration of 10–9. The pour method
was employed to cultivate bacteria, and duplicates of the 1 mL dilutions (10–6
and 10–7) were plated on sterile Petri plates. Further media was poured onto
the Petri plates, homogenized, and then incubated at 37 °C for 48 h. The following
equation was used to calculate the total microbial count:
𝑁=∑ [(1×𝑛1)+(0.1×𝑛2)+...]×(𝑑)
Where,
N = number of colonies per mL/g,
∑C = total number of colonies counted,
n1 = number of cups in the first dilution,
n2 = number of cups in the second dilution,
d = first dilution
calculated [17].
2.11.
Physicochemical attributes of coriander leaves
2.11.1.
Percent Weight Loss for coriander leaves
Selected coriander leaves (three
samples per repetition) for control and freshness keeper treatments were
individually weighed with a calibrated analytical balance on each withdrawal.
On a fresh weight basis, the overall weight loss during storage intervals was
calculated using the following equation:
% weight loss = initial weight − final
weight/initial weight ×
100
Where, % weight loss = percentage of
weight loss and weight of initial sample and final sample are expressed in grams
[17].
2.11.2.
pH
During the storage period, the pH of
fresh coriander leaves was measured by using a Hanna Instruments temperature
bench pH meter HI-3222-02 with 0.001% precision. The material was mashed and
dissolved in 10 mL of distilled water using a total of 1 g. Three measurements
were taken for each treatment [18].
2.11.3.
Titratable acidity
The
titratable acidity (TTA) was measured with some modifications. A 10 mL of
homogenate was mixed with 0.3 mL of 1% phenolphthalein in 95% ethanol and
titrated with 0.1 N sodium hydroxide to a permanent pink color (pH 8.1).
The TTA was calculated using Eq. (2). A
conversion factor of 0.28 was selected based on linoleic acid, a predominant
acid in avocados [18].
TTA= (0.1×𝑁𝑎𝑂𝐻×0.28×1000)
Where,
TTA is the titratable acid, 0.1 is 0.1 mol of NaOH [N],
NaOH is the amount of NaOH added [mL],
0.28 is the conversion factor and S is the juice sample
[mL].
2.12. Sensory
evaluation
A sensory assessment of coriander was done using a structured 9-point hedonic scale, where 9 was scaled as strongly like, and 1 was strongly disliked. The participants of the sensory group were 10 potential consumers who were unbiased individuals from the Faculty of Food Science and Nutrition, B.Z. University, Multan. Individuals on the sensory panel were asked to rate their degree of liking color, odor, texture, and overall palatability on the sensory assessment form Sharma et al. [19].
2.13. Statistical analysis
Analyses
were performed in triplicate. The data was analyzed by analysis of variance
(ANOVA) using SPSS (Version 17.0. Inc., Chicago, USA) statistical program.
Duncan’s multiple range tests were carried out to test any significant
differences among the treatments employed. Significant levels were defined at P
≤ 0.05.
3. Results and discussion
3.1. Effect of
physical properties of sorghum starch film incorporated neem extract
Table 1 showed the result of different properties of starch based film
incorporated with various concentrations of neem extract.
3.1.1 Film
Thickness Measurement
Spoiled sorghum starch based film incorporated with various concentrations of neem extract are shown in Table 1. Film thickness measurement is determined by the millimeter-based thickness of each film sample (mm). The samples vary in thickness, with F0 having the least thickness (0.119 mm) and F1 having the maximum thickness (0.231 mm). F7.5% and F15% have values for thickness of 0.198 mm and 0.201 mm respectively. A thicker film might be able to withstand mass transfer effectively. On the other hand, adding more polymers could make the film thicker by taking up more volume. Our research findings provided support for this concept. The film thickness increased dramatically as the active agent i.e. neem extract was incorporated [20].
Table 1. Effect of physical properties of sorghum starch film incorporated neem extracta
|
Film thickness measurement (mm) |
Film moisture analysis |
Film solubility |
Water vapor permeability ( g/m2/h) |
|
|
F0 |
0.119±0.10a |
24.12±0.26c |
62.12±0.11d |
784.12±0.11b |
|
F1 |
0.231±0.01d |
39.12±0.10d |
52.19±0.10c |
782.19±0.10b |
|
F7.5% |
0.198±0.02c |
10.08±0.10a |
26.36±0.10b |
542.12±0.10a |
|
F15% |
0.201±0.01b |
09.90±0.10b |
22.82±0.01a |
540.02±0.01a |
|
aAll values are means of triplicate
determinations. Means within a column with different superscripts are
significantly different at P < 0.05. Abbreviations: F0: Sorghum
Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%:
Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%:
Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. |
||||
3.1.2 Film moisture analysis
Moisture content in the films shows a notable variation, with F1 having the highest moisture content (39.12), significantly more than the other samples. F0 has moderate moisture content (24.12), whereas F7.5% and F15% have the least moisture contents (10.08 and 09.90, respectively). The presence of hydrophilic additives or a reduced degree of cross-linking may be indicated by the maximum moisture content, which increases the material's susceptibility to absorbing moisture from the environment, whereas the least indicated a composition contained significant hydrophobic components, minimizing moisture absorption [20].
3.1.3 Film solubility
Solubility in water is an important property of starch based films. Solubility trends show a descending order from F0 (62.12±0.11) to F15% (22.82±0.01). The F0 film exhibited the highest solubility, while F15% has the lowest. This indicated that as the percentage increased, solubility decreased which could be due to increased cross-linking or other structural changes in the film composition. The decrease in solubility signifies that the film has stabilized and is now less susceptible to deterioration triggered by water. This can be useful in situations where little resistance to dissolution is needed, like in films or coatings that must tolerate humid conditions.
3.1.4 Water vapor permeability
The water vapor permeability values of F0 and F1 are comparable at 784.12 and 782.19, respectively. However, permeability values for F15% and F7.5% are lower (540.02 and 542.12, respectively), indicating that permeability to water vapor may be decreased when a component's proportion is increased.
The WVP of all modified sorghum starch films tends to decrease except F0 (784.12 g/m2/h). Water vapor permeability is among the most important parameters of edible films since it determines the ability of packaging film to prevent or facilitate moisture uptake from the immediate atmosphere to the food. The WVP could be used to assess the water loss or gain by the food packed in the particular film.
3.2. Effect of Color Profile of sorghum starch film incorporated neem extract
Biodegradable films made from sorghum starch have been examined for CIELAB colour space, which is a standardized color model as shown in Table 2, the L* parameter shows significant (p < 0.05) variation among the samples. The F7.5% sample has the highest L* value (445.2) indicating it is much lighter compared to other films. The F0 sample has the least L* value which is 154.1. The F1 sample is lighter than the F0 and F15% samples but significantly darker than the F7.5% sample. This could be due to the effect of spoilage and the addition of neem extract affecting the transparency or opacity of the films. While all samples have positive a* values. The F7.5% sample has the highest a* value (1.59). The F0 (1.32) and F15% (1.35) samples have similar a* values suggesting that the red color is more influenced by spoilage and less by the neem extract at higher concentrations. The b* values are positive for all samples. The F7.5% sample has the highest b* value (89.0) indicating the most pronounced b*. The F] sample shows a higher value (67) compared to the F0 and F15% samples. The b* seems to increase with spoilage and with the addition of neem extract up to 7.5%. According to the results, the color profile of biodegradable films made from sorghum starch changes significantly with spoilage and the addition of neem extract. The addition of neem extract further enhances these changes, particularly at a concentration of 7.5%. The higher concentration of neem extract (15%) results in a color profile similar to the non-spoiled film. One study showed that at the color profile of biodegradable films made from sorghum starch and found that adding neem extract at a concentration of 7.5% makes the best results. Overall, the incorporation of 7.5% neem extract into sorghum starch films improves their color properties and likely enhances their biodegradability, making them a superior choice for environmentally friendly applications. Previous studies showed that the opacity and color parameters, such as lightness, yellowness, and redness, play a crucial role in food packaging. The capacity to visually examine the food via films is a significant feature that affects the acceptability of the packaging material and the marketability of the wrapped food products [21]. The appearance and reception of products by consumers are significantly impacted by packaging films. When assessing the impact of films on product appeal and consumer acceptability, the instrumental color profile assumes critical significance [22]. EdR, Z. (2014) reported in their research that a* color parameter exhibited a decrease as the concentration of proteins increased [23]. As the protein concentrations increased, the b* parameter of the films exhibited an intensified yellow hue. Transparent films are distinguished by their exceptionally low opacity values. The naturally occurring color of consumable films could potentially be altered through the incorporation of essential oils.
Table 2. Effect of color Profile of sorghum starch film incorporated neem extracta
a* | L* | b* | |
F0 | 54.12±0.01c | 1.01±0.65a | 11.90±2.74ab |
F1 | 78.01±0.01d | 2.31±0.65c | 12.0±2.74a |
F7.5% | 38.85±0.00b | 1.38±0.05b | 14.70±0.024b |
F15% | 26.07±0.01a | 2.57±0.09c | 23.61±0.02c |
aAll values are means of triplicate determinations. Means within a column with different superscripts are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. | |||
3.3. Effect of mechanical testing of sorghum starch film incorporated neem extract
3.3.1 Tensile strength
The tensile strength decreases as we move from F0 to F15%. This suggests that the material becomes progressively weaker with the increasing percentage of the modifying agent or treatment applied to F7.5% and F15%. The F0 and F1 samples have similar tensile strengths, indicating minimal impact of the modification or treatment up to this level.
3.3.2 Percent elongation
According to Table 3, a drop in tensile strength is observed from F0 to F15%. This is due to the fact that when more of the modifying agent or treatment is given to F7.5% and F15%, the material got progressively weaker. The tensile strengths of the F0 and F1 samples are comparable, suggesting that the alteration or treatment had no effect up to this point. While the % elongation increased somewhat, the tensile strength stayed almost the same, indicating that the alteration or treatment at this level only slightly improved ductility without sacrificing strength. Moreover, there is a discernible rise in elongation and a considerable drop in tensile strength. This suggested a trade-off between the material's strength and ductility.
Table 3. Effect of mechanical testing of sorghum starch film incorporated neem extracta
Samples | Tensile strength (MPa) | Percent elongation (%) |
F0 | 4.52±0.01c | 1.45±0.15a |
F1 | 4.56±0.01c | 1.62±0.61b |
F7.5% | 4.12±0.01b | 2.45±0.62d |
F15% | 3.35±0.00a | 2.16±0.29c |
aAll values are means of triplicate determinations. Means within a column with different superscripts are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. | ||
3.4. Effect of biodegradable test of sorghum starch film incorporated neem extract
3.4.1 Weight loss of sorghum starch films during composite test
The biodegradability of spoiled sorghum starch films and their composites was assessed by monitoring the weight loss over a 30-day period. The samples included pure sorghum starch film (F0), spoiled sorghum starch film (F1), and spoiled sorghum starch films with 7.5% and 15% neem extract (F7.5% and F15%, respectively). The results shown in Table 4 demonstrate that weight loss of F0 significantly (p < 0.05) decreases over the 30 days. The pure sorghum starch film has and initial weight loss of 15.01% on 1 day. The initially occurring loss may be attributed to the evaporation of moisture or little degradation of the surface. However, for the next 30 days, the amount of weight lost is insignificant, with a mere 4.90% on the 15th day and 2.12% by the 30th day. These findings suggested that the F0 film has low biodegradability as the weight loss percentage of F0 decreases over the 30-day period.
Table 4. Effect of biodegradable test of sorghum starch film incorporated neem extract
Samples | Percent weight loss of films | ||
| 1 day | 15th day | 30th day |
F0 | 15.01±0.03a,3 | 4.90±0.03a,2 | 02.12±0.00a,1 |
F1 | 10.09±0.01a,1 | 80.02±0.05b,2 | 90.98±0.74b,3 |
F7.5% | 20.09±0.01b,1 | 84.78±0.15c,2 | 98.78±0.14c,3 |
F15% | 40.98±0.01c,1 | 88.89±0.00d,2 | 99.09±0.05c,3 |
aAll values are means of triplicate determinations. Different lower case letters within a column and different numbers within a row are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. | |||
The spoiled sorghum starch film (F1) showed a weight loss of 10.09% on day 1, slightly lower than F0. However, the weight loss significantly (p < 0.05) increases to 80.02% by the 15th day and reaches 90.98% by the 30th day. F1 demonstrated substantial weight loss, specifically after the 15-day period, which suggested a highly rapid rate of biodegradation. F1 has experienced a significant decrease in weight by the 30th day, indicating the biodegradability of the sorghum starch film.
The biodegradability of neem extract-infused sorghum starch films, specifically F7.5% (7.5% neem extract) and F15% (15% neem extract) was analyzed over a 30-day period. The initial weight loss (1 day) for both F7.5% and F15% films is higher than that of pure sorghum starch films i.e. 20.09% and 40.98%. The greater initial weight loss in the F15% film suggested that a higher concentration of neem extract leads to more immediate degradation or solubilization. By the 15th day, both F7.5% (84.78%) and F15% (88.89%) films exhibited significant weight loss with F15% showing a slightly higher degradation rate. This means that neem extract speeds up the biodegradation process. After 30 days, both films approached complete degradation with F15% (99.09%) showing marginally higher weight loss. The near-total weight loss confirmed the highly biodegradable nature of these films. The neem extract concentrations increased the biodegradability of sorghum starch films by a substantial margin. The rate of degradation for F15% films is marginally higher than that of F7.5% films. Both films F15% and F7.5% demonstrated nearly total biodegradation after 30 days, although F15% experienced a slightly greater ultimate weight loss. The results suggested that the incorporation of neem extract into sorghum starch films improved their biodegradability. The F7.5% and F15% films showed rapid and nearly complete degradation within 30 days indicating their potential for environmentally friendly applications requiring quick biodegradation. These films could particularly be useful in agricultural and packaging applications where fast break down into non-toxic components is beneficial. The data suggested that higher neem extract concentrations (15%) slightly enhanced the degradation rate, the difference between 7.5% and 15% is relatively insignificant over the entire 30-day period. This indicated that even lower concentrations of neem extract could be effective in achieving high biodegradability, which could be advantageous from a cost and material efficiency perspective. Previous studies showed that biodegradable films effectively minimize weight loss in comparison to traditional synthetic coatings, indicating improved preservation of food goods. Ensuring the quality and prolonging the shelf-life of perishable items is of utmost importance. These films not only offered sufficient protection but also extended the shelf-life of food goods. The study highlighted the significance of utilizing biodegradable materials for environmental sustainability and food preservation [24]. The weight loss percentage of PVA/CS biodegradable films, after being buried in soil for 30 days, was found to be at least 60%. When compared to the food packaging now available on the market, PVA/CS biodegradable films have the ability to disintegrate rapidly [25]. The rates of biodegradation of the film were determined by measuring the amount of weight loss at various time intervals [26]. The biodegradability of materials mostly relies on the levels of humidity and the chemical composition. The film exhibited the greatest percentage of weight reduction after a period of 28 days, maybe attributed to the hydrophilic properties of chitosan. It may be inferred that the reduction in weight of the starch in the film samples is mostly due to the biodegradation of starch, rather than solely the processes of erosion, weathering, or leaching. Throughout the whole biodegradation test, it was observed that the weight loss of starch cannot be solely attributed to erosion, weathering, or leaching. The weight loss might be mostly attributed to the biodegradation of starch [27].
3.5. Shelf life assessment of composite coriander leaves wrapped with films for 120 days
3.5.1 Total chlorophyll content (mg/100 g)
The table presents the shelf life assessment of total chlorophyll content (measured in mg/100g) for four different samples (F0, F1, F7.5%, and F15%) over a period of 120 days. The data is recorded at 0, 30, 60, 90, and 120 days. All samples start with a total chlorophyll content of 2.1 mg/100 g. F0 (Control Sample): Chlorophyll content decreases gradually from 2.1 mg/100 g at 0 days to 0.9 mg/100 g at 120 days. The decline is relatively steady with statistically significant differences p <0.05 noted over time (indicated by different letters and numbers). F sample drops sharply from 2.1 mg/100g to 0.6 mg/100 g by day 30. By day 60, the chlorophyll content is almost negligible (0.2 mg/100 g), and by day 90, it was zero. F7.5%, decreases slowly compared to F0 and F1, with a slight drop to 2.0 mg/100 g by day 30. Maintains relatively high chlorophyll content even by day 120 (1.7 mg/100 g). F15% showed similar trends to F7.5%, with chlorophyll content reducing slightly to 2.0 mg/100 g by day 30. Ends with a chlorophyll content of 1.6 mg/100 g on day 120. The control sample (F0) showed a gradual decline in chlorophyll content, reflecting natural degradation over time. F1 exhibited the fastest degradation, with chlorophyll content nearly depleted by day 60. This suggests that the additive or treatment used in F1 was not effective in preserving chlorophyll. Both F7.5% and F15% samples had higher chlorophyll levels over the 120-day period compared to F0 and F1. This indicates that the additives used at 7.5% and 15% concentrations were effective in slowing down chlorophyll degradation. The additive at 7.5% and 15% concentrations showed significant efficacy in preserving chlorophyll content. However, there is little difference between the two concentrations in the rate of decline from day 30 to day 120, suggesting that the 7.5% concentration might be sufficient for practical purposes. The rapid decline in F1 could be due to an inappropriate additive or concentration that accelerates chlorophyll degradation rather than inhibiting it.
Table 5. Shelf Life Assessment of composite coriander leaves wrapped with films for 120 days
Samples | Shelf life assessment | ||||
Total chlorophyll content (mg/100g) | |||||
0 day | 30th day | 60th day | 90th day | 120th day | |
F0 | 2.1±0.01a,5 | 1.9±0.02b,4 | 1.5±0.02b,3 | 1.2±0.02b,2 | 0.9±0.02b,1 |
F1 | 2.1±0.01a,4 | 0.6±0.02a,3 | 0.2±0.02a,2 | 0.0±0.02a,1 | 0.0±0.00a,1 |
F7.5% | 2.1±0.01a,4 | 2.0±0.02c,4 | 1.9±0.02c,3 | 1.89±0.02c,2 | 1.7±0.02d,1 |
F15% | 2.1±0.01a,3 | 2.0±0.02c,3 | 1.9±0.02c,2 | 1.81±0.02c,2 | 1.6±0.02c,1 |
| Microbial load | ||||
| 0 day | 30th day | 60th day | 90th day | 120th day |
F0 | 3.81±0.01a,1 | 4.01±0.01b,2 | 4.99±0.01c,3 | 5.91±0.01c,4 | 6.11±0.01b,5 |
F1 | 3.81±0.01a,1 | 8.91±0.01c,2 | 17.81±0.01d,3 | 24.81±0.01d,4 | 37.10±0.01c,5 |
F7.5% | 3.81±0.01a,1 | 3.81±0.01a,1 | 3.92±0.01b,2 | 3.92±0.01b,2 | 3.99±0.01a,3 |
F15% | 3.81±0.01a,1 | 3.83±0.01a,1 | 3.86±0.01a,1 | 3.88±0.01a,1 | 3.89±0.01a,1 |
aAll values are means of triplicate determinations. Different lower case letters within a column and different numbers within a row are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. | |||||
3.5.2 Microbial load
Table 5 presents the microbial load (log CFU/g) of different formulations (F0, F1, F7.5%, and F15%) over a storage period of 120 days. This discussion analyzes the changes in microbial load for each formulation over time and their implications. At day 0, all formulations (F0, F1, F7.5%, and F15%) had the same initial microbial load of 3.81 ± 0.01 log CFU/g, indicating a consistent starting point for all samples. The microbial load in the control sample (F0) gradually increased over time. Starting from 3.81 ± 0.01 log CFU/g at day 0, it rose to 4.01 ± 0.01 log CFU/g by the 30th day and continued to increase, reaching 6.11 ± 0.01 log CFU/g by the 120th day. This steady increase, marked by significant differences at each time point (denoted by different superscript letters and numbers), indicates the absence of any antimicrobial agents in the control formulation. Formulation F1 exhibited a dramatic increase in microbial load. From an initial load of 3.81 ± 0.01 log CFU/g, it surged to 8.91 ± 0.01 log CFU/g by the 30th day and continued to rise sharply, reaching 37.10 ± 0.01 log CFU/g at the 120th day. Each time point shows a significantly higher microbial load than the previous one, suggesting that F1 contains components that either lack antimicrobial properties or promote microbial growth. The microbial load in the F7.5% formulation remained relatively stable compared to F0 and F1. Starting from 3.81 ± 0.01 log CFU/g at day 0, the load remained unchanged at day 30 and showed only slight increases to 3.92 ± 0.01 log CFU/g by day 60 and day 90, and to 3.99 ± 0.01 log CFU/g by day 120. Significant differences were observed only at specific intervals, indicating that the 7.5% formulation has a mild inhibitory effect on microbial growth over the storage period. The microbial load in the F7.5% formulation remained relatively stable compared to F0 and F1. Starting from 3.81 ± 0.01 log CFU/g at day 0, the load remained unchanged at day 30 and showed only slight increases to 3.92 ± 0.01 log CFU/g by day 60 and day 90, and to 3.99 ± 0.01 log CFU/g by day 120. Significant differences were observed only at specific intervals, indicating that the 7.5% formulation has a mild inhibitory effect on microbial growth over the storage period. The results clearly showed that formulations F7.5% and F15% have significant antimicrobial effects compared to F0 and F1. While the control (F0) and F1 formulations allowed substantial microbial growth, F7.5% and particularly F15% formulations effectively inhibited microbial proliferation.
3.6. Physicochemical attributes of composite coriander leaves wrapped with films for 120 days.
3.6.1 Moisture content
The results obtained on the biodegradable films (Table 6) showed a significant (p < 0.05) impact on moisture content. Moisture content is critical parameter. In F0 the moisture content gradually rises from 88.01% on 0 day to 91.09% on 120th day. The slow increase seen suggested the possibility of moisture being absorbed from the environment which is a common characteristic of films made from starch. In F1 film, the moisture content increased rapidly from 88.09% on day 0 to a considerable 100.18% on 120th day. The significant rise particularly within the 60th to 120th day timeframe indicated an enhanced uptake of moisture potentially resulting from alterations in the film's composition induced by spoilage. In F7.5% results showed a more stable moisture content, starting at 88.09% and increasing slightly to 89.24% by 120th day. The stability suggested that the addition of neem extract may have a protective impact decreasing the film's sensitivity to absorbing moisture. In F15% the moisture content decreases from 88.98% at day 0 to 86.01% by day 120. The decline implies that a greater concentration of neem extract may cause a drying effect, resulting in a gradual reduction of the film's moisture content.
Table 6. Physicochemical attributes of composite coriander leaves wrapped with films for 120 days.
Samples | Physicochemical attributes | ||||
Moisture content | |||||
0 day | 30th day | 60th day | 90th day | 120th day | |
F0 | 88.01±0.03a,1 | 89.90±0.03c,2 | 89.12±0.00b,2 | 90.98±0.00c,3 | 91.09±0.00c,3 |
F1 | 88.09±0.01a,1 | 90.02±0.05d,2 | 90.18±0.74c,2 | 90.74±0.00c,2 | 100.18±0.00d,3 |
F7.5% | 88.09±0.01a,1 | 88.24±0.15b,1 | 88.58±0.14b,1 | 88.94±0.00b,1 | 89.24±0.00b,2 |
F15% | 88.98±0.01a,3 | 87.79±0.00a,2 | 87.09±0.05a,2 | 86.2±0.00a,1 | 86.01±0.00a,1 |
| pH | ||||
| 0 day | 30th day | 60th day | 90th day | 120th day |
F0 | 6.10±0.01a,4 | 5.90±0.03b,3 | 5.80±0.00b,3 | 4.71±0.00a,2 | 4.01±0.00b,1 |
F1 | 6.10±0.01a,4 | 4.90±0.05a,3 | 4.80±0.74a,2 | 4.29±0.00a,2 | 3.99±0.01a,1 |
F7.5% | 6.10±0.01a,1 | 6.10 ±0.15c,1 | 6.20±0.14c,2 | 6.21±0.00c,2 | 6.22±0.01d,2 |
F15% | 6.10±0.01a,2 | 6.10±0.00c,2 | 6.19±0.05c,4 | 6.12±0.00b,3 | 5.90±0.00c,1 |
| Titratable Acidity | ||||
| 0 day | 30th day | 60th day | 90th day | 120th day |
F0 | 0.063±0.03a,1 | 0.061±0.03a,1 | 0.072±0.00c,2 | 0.081±0.00b,3 | 0.098±0.00b,4 |
F1 | 0.063±0.01a,1 | 0.213±0.05c,2 | 0.288±0.74d,2 | 0.668±0.00c.3 | 0.987±0.00c,4 |
F7.5% | 0.063±0.01a,3 | 0.063±0.15b,3 | 0.061±0.14b,2 | 0.059±0.00a,1 | 0.059±0.00a,1 |
F15% | 0.063±0.01a,2 | 0.060±0.00a,2 | 0.059±0.05a,1 | 0.059±0.00a,1 | 0.059±0.00a,1 |
aAll values are means of triplicate determinations. Different lower case letters within a column and different numbers within a row are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract. | |||||
Both F0 and F1 are highly reactive to an increase in moisture content which may influence their biodegradability. Higher moisture levels might cause a decrease in tensile strength and expedited biodegradation. Films containing neem extract at concentrations of 7.5% and 15% exhibit a higher degree of moisture control. The formulation containing 15% neem extract specifically maintains reduced moisture levels which is advantageous for applications that necessitate more resilient and long-lasting coatings. Neem extract enhances moisture resistance with higher concentrations providing greater stability.
Previous results showed the binding of water may be influenced by the density and regularity of the packing of the polysaccharide chains [28]. Biodegradable films for the food industry may interact with goods with varying moisture content. Since starch is a hydrophilic polymer, these packaging materials must be tested in multiple equilibrium relative humidity conditions to determine their suitability as food packaging [29].
3.6.2 pH
The pH of biodegradable films plays an important role in their functionality, stability, and interaction with the environment. Results showed in Table 6 significant (p < 0.05) decrease in pH of F0 and F1 pH from 6.1 to 4.0 in 120 days which indicated the increasing acidity over time. In F7.5%, pH remained relatively stable starting at 6.1 similar to F15% showing minor fluctuations by day 120. This firmness indicated that neem extract effectively inhibited microbial activity and prevented acidification. This stability can lead to the production of more long-lasting films with an extended shelf life making them appropriate for applications that demand regular pH levels [30].
3.6.3 Titratable acidity (TA)
The results of titratable acidity Table 6 indicated that at 0 day samples had 0.063 TA, which increased significantly (p<0.05) from 0.063 at day 0 to 0.987 by 120th day suggested a slow buildup of acidic compounds likely due to minor microbial activity. While F7.5% and F15% remain stable at around 0.059 showing no significant change over the 120 days. The higher concentration of neem extract provides robust protection against spoilage preventing any increase in acidity.
3.7. Sensory evaluation
Table 7 demonstrated the sensory analysis of coriander leaves wrapped through films across the period of 120 days at specific intervals (0 day, 30th day, 60th day, 90th day and 120th day).
Table 7. Sensory Evaluation of composite coriander leaves wrapped with films for 120 days.
Samples | Parameters | Physicochemical attributes | ||||
0 day | 30th day | 60th day | 90th day | 120th day | ||
F0 | Color | 10±0.00a,4 | 7.00±0.00b,3 | 6.60±0.57b,2 | 1.00±0.57a,1 | 0 |
F1 | 10±0.00a,2 | 1.66±0.57a,1 | 0.66±0.57a,1 | 0 | 0 | |
F7.5% | 10±0.00a,3 | 8.33±0.57c,2 | 8.00±0.57c,2 | 6.33±0.57b,1 | 0 | |
F15% | 10±0.00a,3 | 8.66±0.57c,2 | 8.66±0.57c,2 | 6.66±0.57b,1 | 0 | |
F0 | Texture | 10±0.00a,3 | 8.00±0.57b,2 | 5.33±0.577a,1 | 0 | 0 |
F1 | 10±0.00a,2 | 4.00±0.57a,1 | 0 | 0 | 0 | |
F7.5% | 10±0.00a,3 | 8.66±0.57c,2 | 8.00±0.577b,2 | 6.33±0.577a,1 | 0 | |
F15% | 10±0.00a,3 | 9.00±0.57c,2 | 8.66±0.577b,2 | 7.00±0.577b,1 | 0 | |
F0 | Odor | 10±0.00a,2 | 9.00±0.00b,1 | 0 | 0 | 0 |
F1 | 10±0.00a,2 | 1.67±0.57a,1 | 0 | 0 | 0 | |
F7.5% | 10±0.00a,3 | 9.00±0.57b,2 | 5.00±0.577a,1 | 0 | 0 | |
F15% | 10±0.00a,3 | 9.00±0.57b,2 | 5.33±0.577a,1 | 0 | 0 | |
F0 | Overall Acceptability | 10±0.00a,3 | 6.00±0.00b,2 | 4.60±0.01a,1 | 0 | 0 |
F1 | 10±0.00a,2 | 2.00±0.57a,1 | 0 | 0 | 0 | |
F7.5% | 10±0.00a,2 | 8.33±0.57c,1 | 8.33±0.57b,1 | 0 | 0 | |
F15% | 10±0.00a,2 | 9.00±0.57c,1 | 9.00±0.57b,1 | 0 | 0 | |
aAll values are means of triplicate determinations. Different lower case letters within a column and different numbers within a row are significantly different at P < 0.05. Abbreviations: F0: Sorghum Starch Film, F1: Spoiled Sorghum Starch Film, F7.5%: Spoiled Sorghum Starch Film incorporated with 7.5% Neem Extract, F15%: Spoiled Sorghum Starch Film incorporated with 15% Neem Extract.
3.7.1 Color
The initial score was 10 on zero day but over the time color degraded significantly. Coriander leaves wrapped by Fo and F1 samples showed drastic decrease. At the beginning, F0 received high but it reached 1 by the 90th day and disappeared by the 120th day. F1 sample observed decline in color much faster from 10 dropping drastically by the 30th day and reached zero after the 60th day. This could be due to the higher concentration of microorganism was already established in the film, which causes deterioration to the coriander leaves. In contrast, F7.5% and F15% had better color stability.
3.7.2 Texture
The texture, as shown in Table 7 also declined over time. Sample F1 experienced a sharper declined falling to zero on the 60th day. Whereas better texture retention of coriander leaves was observed in F7.5% and F15%. Even by the 90th day, they maintained relatively high texture scores.
3.7.3 Odor
The evaluation of odor also revealed variation with the treatments being applied to coriander leaves, as shown in Table 7. In the beginning, all films had high odor retention but, afterwards, the odor of coriander leaves within samples of F0 and F1 showed a rapid decrease by the 60th day. Whereas, samples of F7.5% and F15% had relatively higher odor scores till the 60th day. Thus, coriander leaves wrapped within the samples of F7.5% and F15% could help in maintaining the aroma of coriander during storage.
3.7.4 Overall acceptability
Finally, the overall acceptability of coriander leaves was also fairly high and followed the same trend as individual parameters. F0 showed a significant decline by the 90th day reaching zero on the 120th day. Whereas F1 dropped to zero by 60 days. Coriander leaves wrapped within the sample of F7.5% and F15%. Maintained good acceptability through 60 days but by 90 days they also declined.
Therefore, the findings of this study suggested that coriander leaves stored in concentrations of F7.5% and F15% films yielded improved sensory characteristics, including color, texture, odor and overall acceptability than those in low films F1. This data implies that the density of the packaging film should be enhanced in order to get the neem extract concentration that could enhance the shelf life of coriander leaves stored at room temperature.
4. Conclusions
The rate of spoilage for F15% films is marginally higher than that of F7.5% films. Both films F15% and F7.5% are totally biodegradable after 30 days, although F15% experienced a slightly greater ultimate weight loss. F7.5% showed the best results of the color profile and enhanced the biodegradability of the films that contained neem extract at concentrations of 7.5% and 15%, showed a higher degree of moisture control. The pH remains stable at F7.5% and F15% stable pH extends the shelf life. In titration acidity F7.5% and F15% remained stable at 0.059. The study indicated that the additives used in F7.5% and F15% samples are effective in preserving chlorophyll content over an extended period. For applications requiring prolonged shelf life and retention of chlorophyll, using a 7.5% concentration of the additive could be a cost-effective and efficient approach. Further studies could explore the exact mechanism by which these additives preserved chlorophyll and whether higher concentrations would yield any additional benefits. These findings are crucial for food preservation strategies. The effectiveness of the 15% formulation in inhibiting microbial growth suggested it could be used to enhance the shelf life and safety of food products. Further research could explore the specific components and mechanisms behind the antimicrobial effects observed in the F7.5% and F15% formulations, as well as their applications in various food systems.
Authors’ contributions
Supervision, conception and design, D.S.S.; methodology, M.F.J.K.; investigation, R.A.M.; analysis and validation, M.A.J.; wrote the manuscript, M.J.U.D.; statistical analysis and reviewed the manuscript, H.C.
Acknowledgements
The authors don't have anything to acknowledge.
Funding
This research received no external funding
Availability of data and materials
All data will be made available on request according to the journal policy
Conflicts of interest
The authors declare no conflict of interest
References
1.
Bakari, H.; Djomdi, Ruben, Z.F.; Roger, D.D.; Cedric, D.;
Guillaume, P.; Pascal, D.; Philippe, M.; Gwendoline, C. Sorghum
(Sorghum bicolor L. Moench) and its
main parts (by-products) as promising sustainable sources of value-added
ingredients. Waste Biomass Valor. 2023, 14(4),
1023-1044. https://doi.org/10.1007/s12649-022-01992-7
2.
Virmani,
S.; Rao, M.; Srivastava, K. Approaches to the management of Vertisols in the
semi-arid tropics: The ICRISAT experience, 1989. http://oar.icrisat.org/id/eprint/4431.
3.
Okparavero,
N.F.; Grace, O.O.; Rukayat, Q.; Jimoh, O.; Ishola, T.; Okunlade, A.; Haruna, P.;
Isaac, A.; Akande, E. Effective Storage structures
for preservation of stored grains in Nigeria: A review. Ceylon J, Sci. 2024,
53(1), 139-147.
4.
Ackerman,
A.; Wenndt, A.; Boyles, R.; The sorghum grain mold disease complex: Pathogens,
host responses, and the bioactive metabolites at play. Front. Plant Sci. 2021,
12 660171.
https://doi.org/10.3389/fpls.2021.660171.
5.
Kange, A.M.; Cheruiyot, E.K.; Ogendo, J.O.; Arama, P.F.; Effect of sorghum (Sorghum bicolor L. Moench) grain conditions on occurrence of
mycotoxin-producing fungi. Agri. Food Secur. 2015, 4, 1-8. https://doi.org/10.1186/s40066-015-0034-4.
6.
Purnama,
I.; Montesqrit, H. The effect of neem
leaves powder (Azadiractha indica A.
Juss) and storage time to rejected corn quality. Int. J. Env.
Agric. Biotechnol. 2019,
4(6). https://doi.org/10.22161/ijeab.46.39.
7.
Elvira,
N.D.; Wuryandari, Y. Utilization of neem leaves as a biological pesticide for
the control of anthrachnose diseases in dragons (Hylocereus sp.). Nusantara Sci.
Technol. Proc. 2023, 36-40. https://doi.org/10.11594/nstp.2023.3109.
8.
Demirak,
M. Ş.S.; Canpolat, E. Plant-based
bioinsecticides for mosquito control: Impact on insecticide resistance and
disease transmission. Insects. 2022, 13(2), 162. https://doi.org/10.3390/insects13020162.
9.
Davis,
G.; Song, J. Biodegradable packaging based on raw materials from crops and
their impact on waste management. Ind. Crops Prod. 2006, 23(2), 147-161. https://doi.org/10.1016/j.indcrop.2005.05.004.
10.
Mohanty,
A.K.; Misra, M.; Hinrichsen, G. Biofibres, biodegradable polymers and
biocomposites: An overview. Macromol. Materials Engin. 2000, 276(1), 1-24. https://doi.org/10.1002/(sici)1439-2054(20000301)276:1%3C1::aid-mame1%3E3.0.co;2-w.
11.
Bidoia,
E.D.; Montagnolli, R.N. Biodegradation, pollutants and bioremediation
principles. CRC Press, 2021. https://books.google.com.pk/books?id=HPwTEAAAQBAJ.
12.
Mehboob,
S.; Ali, T. M.; Alam, F.; Hasnain, A. Dual modification of native white sorghum
(Sorghum bicolor) starch via acid
hydrolysis and succinylation. LWT-Food Sci. Technol. 2015, 64(1), 459-467. https://doi.org/10.1016/j.lwt.2015.05.012.
13.
Ranjit, R.R.; Sawant, A.R.; Jamge, B. Antimicrobial activity
of Azadirachta indica (Neem) against
pathogenic microorganisms. J.
Acad. Ind. Res. 2014, 3( 7), 327-329.
14.
Pareta,
R.; Edirisinghe, M.J. A novel method for the preparation of starch
films and coatings." Carbohydr. Polym. 2006, 63(3), 425-431. https://doi.org/10.1016/j.carbpol.2005.09.018.
15.
Mehboob,
S.; Ali, T.M.; Sheikh, M.; Hasnain, A. Effects of cross linking and/or
acetylation on sorghum starch and film characteristics. Int. J. Biol. Macromol.
2020, 155, 786-794. https://doi.org/10.1016/j.ijbiomac.2020.03.144.
16.
Nigam,
S.; Das, A.K.; Patidar, M.K. Synthesis, characterization and biodegradation of
bioplastic films produced from Parthenium hysterophorus by incorporating a
plasticizer (PEG600). Env. Challeng. 2021, 5, 100280. https://doi.org//10.1016/j.envc.2021.100280.
17.
Pawde,
S.; Chaudhari, S.R.; Matche, R.S. Active packaging for strawberry and coriander:
A natural extract impregnated paper. ACS Food Sci. Technol. 2024, 4(5),
1166-1178. https://doi.org/10.1016/j.afres.2022.100122.
18.
Tokatlı,
K.; Demirdöven, A. Effects of chitosan edible film coatings on the
physicochemical and microbiological qualities of sweet cherry (Prunus avium L.). Sci. Hor. 2020, 259,
108656. https://doi.org/10.1016/j.scienta.2019.108656.
19.
Sharma,
H.; Sharma, B.D.; Mendiratta, S.; Talukder, S.; Ramasamy, G. Efficacy of
flaxseed flour as bind enhancing agent on the quality of extended restructured
mutton chops. Asian-Aust. J. Animal Sci. 2014, 27(2), 247. https://doi.org/10.5713/ajas.2013.13319.
20.
Alam,
F.; Nawab, A.; Lutfi, Z.; Haider, S.Z. Effect of non‐starch
polysaccharides on the pasting, gel, and gelation properties of taro (Colocasia esculenta) starch. Starch‐Stärke,
2021, 73(1-2), 2000063. https://doi.org/10.1002/star.202000063.
21.
Chatli,
M.K.; Wagh, R.V.; Mehta, N.; Kumar, P.; Malav, O.P. Quality characteristics of
composite biodegradable films for food packaging. Nutr. Food Sci. 2021, 51(2),
205-221. https://doi.org/10.1108/nfs-01-2020-0015.
22.
Ramos,
Ó.L.; Reinas, I.; Silva, S.I.; Fernandes, J.C.; Cerqueira, M.A.; Pereira, R.N.;
Vicente, A.A.; Poças, M.F.; Pintado, M.E.; Malcata, F.X. Effect of whey protein
purity and glycerol content upon physical properties of edible films
manufactured therefrom. Food Hydrocol. 2013, 30(1), 110-122. https://doi.org/10.1016/j.foodhyd.2012.05.001.
23.
Zavareze, E.daR.; Halal, S.L.M. E.;
Marques e Silva, R.; Dias, A.R.G.; Prentice-Hernández, C. Mechanical, barrier and
morphological properties of biodegradable films based on muscle and waste
proteins from the Whitemouth Croaker (Micropogonias
furnieri). J.
Food Process. Preserv. 38, 2014, 1973-1981. https://doi.org/10.1111/jfpp.12173.
24.
Tharanathan,
R. Biodegradable films and composite coatings: past, present and future. Trends
Food Sci. Technol. 2003, 14(3), 71-78. https://doi.org/10.1016/S0924-2244(02)00280-7.
25.
Yu, Z.; Li, B.; Chu, J.; Zhang, P. Silica in situ enhanced PVA/chitosan
biodegradable films for food packages. Carbohyd. Poly. 2018, 184, 214-220. https://doi.org/10.1016/j.carbpol.2017.12.043.
26.
Indumathi,
M.; Rajarajeswari, G. Mahua oil-based polyurethane/chitosan/nano ZnO composite
films for biodegradable food packaging applications. Int. J. Biol. Macromol.
2019, 124, 163-174. https://doi.org/10.1016/j.ijbiomac.2018.11.195.
27.
Wang,
H.; Wei, D.; Zheng, A.; Xiao, H. Soil burial biodegradation of antimicrobial
biodegradable PBAT films. Polymer degradation and stability, 2015, 116, 14-22. https://doi.org/10.1016/j.polymdegradstab.2015.03.007.
28.
Andrade-Mahecha,
M.M.; Tapia-Blácido, D.R.; Menegalli, F.C. Development and optimization of
biodegradable films based on achira flour. Carbohyd. Polym. 2012, 88(2),
449-458. https://doi.org/10.1016/j.carbpol.2011.12.024.
29.
Gómez‐Aldapa, C.A.; Velazquez, G.; Gutierrez, M.C.; Castro‐Rosas, J.; Jiménez‐Regalado, E. J.; Aguirre‐Loredo, R.Y. Characterization of functional
properties of biodegradable films based on starches from different botanical
sources. Starch‐Stärke, 2020, 72(11-12), 1900282. https://doi.org/10.1002/star.201900282.
30.
Veiga‐Santos,
P.; Ditchfield, C.; Tadini, C.C. Development and evaluation of a novel pH
indicator biodegradable film based on cassava starch. J. Appl. Poly. Sci. 2011,
120(2), 1069-79. https://doi.org/10.1002/app.33255.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
The
research investigated the restoration of spoil sorghum by utilizing neem
extract to create biodegradable films. Sorghum is one of the most important cereal
crops that frequently spoils and causes a lot of waste. In an effort to lessen
the waste, the research was to make biodegradable, eco-friendly films that
would increase the worth and cut down on waste. The antibacterial and
antioxidant qualities of neem extract were added to the sorghum-based film
matrix. The resulting films were assessed for biodegradability, antibacterial
effectiveness, and mechanical, thermal, and barrier qualities. The results suggested
that the incorporation of 7.5% neem extract into sorghum starch films improved
their biodegradability and also color properties. F7.5% shows more
stable result of moisture content while F15% causes drying effect
which results in reduction of film moisture content. The pH values in F7.5%
and F15% remain stable indicated that neem extract effectively
inhibits microbial activity and prevents acidification.
Abstract Keywords
Spoiled
sorghum starch, neem extract, biodegradable films, film solubility, water
vapour permeability, mechanical test, shelf life of coriander leaves.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).



